Abstract
Context:
Infections are a major cause of disease and mortality in transplant recipients. Despite the studies conducted in Iran, no comprehensive and general research is available in this area. The present study aimed to determine the frequency of infectious agents in patients after bone marrow transplantation in Iran.Method:
In this systematic review, relevant studies were selected based on type and objective, and data were collected from the articles published in Iran regarding the frequency of infectious agents after bone marrow transplantation in different regions of Iran. The studies were collected using systematic search methods.Results:
In total, 11 studies were identified regarding infectious agents after bone marrow transplantation. Six studies were conducted in Tehran, three studies were performed in Shiraz, and Mashhad and Semnan provinces were the locations of two separate studies. Most of the case studies identified viral agents (54.5%; n = 6), followed by fungal infectious agents (27.3%; n = 3) and bacterial agents (18.2%; n = 2). Gram-positive bacteria (bacterial agents), cytomegalovirus (viral agents), and Candida and Aspergillus (fungi) had the highest frequency after bone marrow transplantation.Conclusions:
According to the results, viral, fungal, and bacterial infectious agents were respectively most frequent in patients receiving bone marrow transplants. Gram-positive bacteria (bacterial agents), cytomegalovirus (viral agents), and Candida and Aspergillus (fungi) had the highest frequency after bone marrow transplantation.Keywords
Bone Marrow Transplantation Infections Systematic Review Regions of Iran
1. Context
Bone marrow transplantation (BMT) is the re-establishment of normal blood cell production through the long-term transplantation of hematopoietic stem cells. This technique is used to replace absent, malignant, or genetically abnormal stem cells or to save the patient from chemotherapy and radiotherapy against bone marrow-destroying agents (1, 2). BMT is used for the treatment of various diseases, such as aplastic anemia (inability to make new tissues), leukemia, bone marrow cancer, immune system deficiency, lymphoid tissue tumors (lymphomas; e.g., lymphadenoma [Hodgkin's disease]), and rigid tumors such as breast cancer and ovarian cancer (3, 4).
Various types of BMT include allogeneic BMT (5), autologous BMT (self-transplantation) (6), and syngeneic BMT (identical twin transplant) (7, 8). BMT could be performed by the allogeneic method (from the donor) or the autologous method (from the patient's own) (9, 10).
Following BMT, numerous complications may occur in the patient, and the frequency of these complications depends on the type of transplantation, age and health status of the patient, and underlying diseases. Some of these complications are caused by the medications used to prepare the patient, which should be continued after transplantation to preserve the transplant. On the other hand, some BMT complications are caused by the immune reactions of the patient after receiving the transplant. These complications include graft rejection, graft reaction against the host, infections, malignancies, drug complications, hemorrhage, and bone marrow suppression (11, 12).
After transplantation, the patient is susceptible to life-threatening infections for two reasons; first, the bone marrow lacks any cells at first, and the patient is unable to cope with various factors. Second, using immunosuppressants to suppress the immune system exposes the patient to infectious diseases (1, 13). In such cases, infection could be prevented by taking measures such as strict adherence to the principles of patient isolation, systemic antibiotic consumption prophylactically, rapidly obtaining cultures from the blood, urine, throat, and stool secretions rapidly in the case of fever, respiratory physiotherapy to prevent atelectasis and the subsequent pneumonia, controlling vital signs accurately and regularly, assessing the specific supplies of the patient, examining the visitors of the patient in terms of infections, cleaning the room and its equipment on a daily basis, and washing the patient’s fruits and vegetables carefully (14, 15).
Infections are a major cause of disease and mortality in transplant recipients. These patients are exposed to primary infections and at the risk of activating latent infections or infections with agents transmitted through the transplanted tissue (16). Previous studies in Iran have investigated infectious agents after BMT. However, no comprehensive and complete studies are available in this regard.
The present study was aimed to determine the frequency of infectious agents after BMT in Iran based on the previous studies in this regard.
2. Method
This systematic review was conducted to identify the studies regarding the frequency of infectious agents after BMT. We searched for the articles published in domestic and foreign journals and reputable in databases such as ISI Web of Science, Cochrane Library, PubMed, MEDLINE, EMBASE, Cochrane, CINAHL, MD Consult, ScienceDirect, Google Scholar, Magiran, Ovid, Irandoc, Scopus, Scientific Information Database (SID), and Index Medicus. The literature search was performed using a systematic search method and various keywords, which were selected based on MESH standards, including Opportunistic Infections, Bone Marrow Transplantation, Viral Infection, Bacterial Infection, Transplantation, and their Persian equivalents.
After identifying and extracting the articles from the databases, their titles and abstracts were investigated by the researchers. The eligibility criteria of the study were available full-text articles regarding infectious agents after BMT and complete information. The exclusion criteria were case reports and studies investigating infections in other types of organ transplantations and their associated diseases.
The selected articles were carefully investigated by the researchers, and the articles relevant to the research subject were selected. Following that, the required data were collected from the articles, including the name of the first author, year of publication, type of study, geographical location of the study, number of patients, type of infection, gender of the patients, and the species of the identified microorganisms. Other data were also extracted and recorded in relevant forms. After investigation, the obtained data were classified by the researchers, analyzed, and interpreted.
3. Results
In total, 48 articles were identified in Persian and English languages, which investigated infectious agents after BMT in Iran. These articles were reviewed by the researchers, and 11 articles that met the eligibility criteria of the study were selected for further review. Figure 1 shows the results of investigating the articles.
Results of article reviews
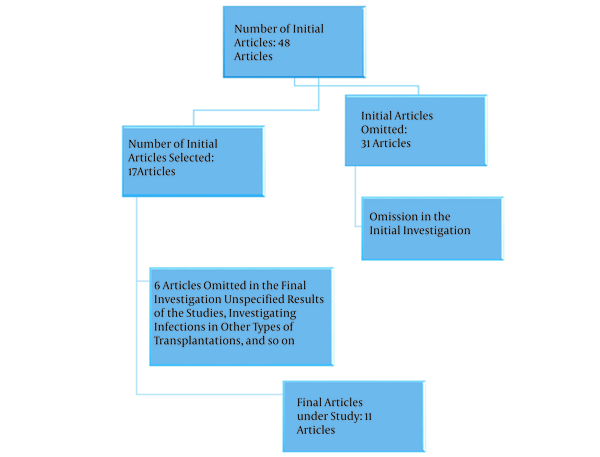
In 11 reviewed studies aiming to identify the infectious agents after BMT performed in Iran, the study by Saderi et al. compared adenovirus secretion in the urine of patients receiving bone marrow before and after transplantation in 2005 in Tehran, Iran. In the mentioned study, 91 patients received sample BMT, and two urine samples were obtained from 72 patients before and four weeks after transplantation. Adenovirus DNA was detected in the urine samples of 39 patients (54.2%) before transplantation, as well as the samples of 37 patients (51.4%) four weeks after transplantation. According to the findings, both urine samples (before and after transplantation) were negative in 21 patients and positive in 25 patients, whereas only the sample before the transplantation (14 patients) and only the sample after transplantation (12 patients) were positive. This investigation showed no increase in adenovirus infection in the patients receiving BMT. Furthermore, no correlation was observed between individual variables and the prevalence of adenovirus in the urine samples (17). In another study, Shirazi et al. investigated bacterial infections in BMT recipients in Tehran (Iran) in 2007, reporting that bacterial infections (especially infection with gram-positive cocci) were still a major issue associated with BMT (18).
The study conducted by Bagheri aimed to identify the infection of DNA viruses and failure in BMT in the south of Iran, and the obtained results indicated the role of pathogenic viral infections in BMT failure in the patients (19). In Shiraz (Iran), the study by Badiee and Alborzi aimed to identify Aspergillus species in patients undergoing BMT. Out of 993 blood samples collected from 82 BMT candidates, 94 were positive DNA samples. Moreover, the mentioned study indicated that Aspergillus infections could be identified in the blood samples of the BMT candidates using polymerase chain reaction (PCR). Early diagnosis of Aspergillus infections by PCR could also decrease patient mortality and reduce the treatment costs of hospitals (20). In another study, Koukhaei et al. assessed the level of the BK virus in the urine of patients undergoing BMT and evaluated its effects on the diagnosis of hemorrhagic cystitis (HC). Although no significant correlation was reported between the high ratio of the virus and the C → G mutation, a higher copy (number) ratio than μL 104 indicated the risk of HC (21).
In the study conducted by Ghiasian and Kord-Bachae in Tehran, opportunistic fungal infections were investigated in patients with blood malignancies and bone marrow transplant recipients. During one year, the researchers examined 32 bone marrow transplant recipients who were hospitalized in Dr. Shariati Hospital in Tehran and had various blood disorders. During the study, nine transplant recipients showed suspected clinical signs of fungal infections (once or twice), and 15 patients developed fungal infections. In a direct experiment and culture, various Candida species were also isolated in 10 patients (66.7%), Geotrichum candidum was detected in one case (6.7%), Cryptococcus albidus was identified in one case, and three unknown yeast species were also detected. All the patients (except two cases) responded to antifungal treatments. In addition, nine cases of the colonization of fungal agents were observed, six of which were observed before BMT, and three cases were reported after the operation. In the mentioned study, the patients received treatment with amphotericin B and nystatin (22).
A comparative study by Hashemi and Shouhani measured the saprophytic fungi found in the space and equipment of blood and oncology research centers and the clinical specimens of transplant recipients in Dr. Shariati Hospital in Tehran. Despite using preventive tools, the patients contracted saprophytic fungal infections from the hospital environment (23). In a similar study conducted by Khandan et al. in Tehran, the frequency of the cytomegalovirus (CMV) antigen was evaluated in the peripheral blood leukocytes of bone marrow transplant recipients. Among 145 patients undergoing BMT for various reasons at different ages, 60 cases had a positive antigen for PP65 (41.4%), out of which 37 cases were male and 23 were female. In those receiving an autologous transplant (15.4%) and in those receiving an allogeneic transplant, the frequency of the antigen was estimated at 47%, indicating the increased ratio of the CMV antigen in the recipients of an allogeneic transplant (24).
In a retrospective study carried out in Tehran by Safayi et al., the researchers investigated infections in patients with acute myeloid leukemia receiving allogeneic BMT, reporting that out of 49 patients, only one case of fever for seven days was observed after receiving the BMT. In the mentioned study, the rate of sepsis was estimated at 6.1%. No fungal infections were observed in the patients, and lungs were the most common site of infection (70%). In addition, the CMV antigen ratio was determined to be 20% during the two-year period after the transplantation. The CMV antigen ratio reported in the mentioned study (20%) was lower than other similar studies (51%), which could be owing to suitable organ donors and recipients and the diminished need for immunosuppressants (25).
In a descriptive, cross-sectional study conducted by Tousi et al., the microbial and neutropenic diet of patients undergoing BMT was evaluated in Mashhad (Iran) in 2018. In the mentioned study, the total counts of bacteria and coliforms were determined, including yeasts and bacteria such as Bacillus cereus. Coliforms were also isolated from the samples with a low count, and no contamination was detected in these samples. As a result, the neutropenic diet used for these patients was fully compliant with the standards of the FDA and the Ministry of Health in terms of foodstuff decomposition (26).
The research performed by Mohammadi et al. aimed to investigate the prevalence of viral infections in BMT candidates in Shiraz (Iran). Among 27 patients, hepatitis B and parvovirus B19 viruses were detected in nine (33%) and seven cases (26%), respectively. In addition, hepatitis C and G viruses were observed in three patients (11%), while the BK virus was detected in none of the cases. The mentioned study indicated the high prevalence of hepatitis B virus and parvovirus B19 in patients with bone marrow suppression, which highlights the importance of these viral infections in these patients (27).
4. Discussion
Among 11 reviewed studies regarding the frequency of infectious agents after BMT, six studies were conducted in Tehran, three were conducted in Shiraz, and two studies were performed in Mashhad and Semnan provinces separately. Most of these studies aimed to identify viral agents and reported the frequency of 54.5% (six cases), while the prevalence of fungal and bacterial infections was estimated at 27.3% (three cases) and 18.2% (two cases), respectively. In the study conducted by Shirazi et al. in Tehran, 52 blood samples and 25 catheter specimens were obtained from 23 BMT candidates with the mean age of 28 years (60% males). In total, 12 bacterial isolates were collected, including eight staphylococcus specimens (66.7%), two Pseudomonas aeruginosa specimens (16.7%), one streptococcus specimen (8.3%), and one Staphylococcus aureus specimen (8.3%). The mentioned study indicated that gram-positive bacteria were the agent of more than 75% of the infections in the patients (18).
In a similar study performed in Spain, gram-positive bacteria were detected in 85% of the cases (staphylococci and streptococci) as the most prevalent bacteria causing infection in BMT candidates (28). In the study by Safayi et al., none of the transplant recipients were infected with pulmonary tuberculosis, and only one of the patients had pulmonary tuberculosis before transplantation, which was treated completely (25).
In various studies, the prevalence of viral infections in BMT recipients has been reported to be 8 - 10% (29, 30). Based on several clinical and laboratory findings, some viral infections have been associated with the pathophysiology of the early temporal bone suppression. In the study by Mohammadi et al., the prevalence of viral infections was evaluated in BMT candidates in Shiraz province (Iran). Among 27 investigated patients, hepatitis B and parvovirus B19 viruses were detected in nine cases (33%) and seven cases (26%), respectively. Hepatitis C and G viruses were also observed in three patients (11%), while the BK virus was identified in none of the cases (27).
CMV could inhibit the hematopoietic process by targeting the functional segments of the bone marrow, thereby leading to infections and the disruption of BMT. Deficiencies in bone marrow stromal function in the early stages halt the process of hematopoiesis and lead to bone marrow aplasia (31). CMV may also alter the function of lateral cells, thereby reducing the production of hematopoietic factors or changing the expression of the adhesion molecules of the cell surface and causing infection in primary hematopoietic cells (32). In another study, 145 patients were examined after BMT, and 60 cases (41.4%) of CMV were identified in 37 male and 23 female patients. In those who had received an autologous transplant (15.4%) and an allogeneic transplant, the frequency of CMV was estimated at 47%, indicating the increased ratio of CMV in the recipients of an allogeneic transplant (24). On the other hand, Safayi et al. reported the lower prevalence of CMV infection in BMT candidates (20%) (25). In a study conducted in Brazil, the prevalence of CMV in BMT candidates was reported to be 51% (33). This discrepancy could be due to the complete compatibility of the transplant donors and recipients, which reduces the need for immunosuppressive treatment.
In another study performed in Iran, CMV was not identified in the samples of BMT candidates (19). In the study by Bagheri, 27 plasma samples were collected during two years, and herpesvirus type 6 and 8 and TT virus 3 (11.1%) were detected in two cases (7.4%) and one patient (3.7%), respectively (19). In other countries, herpesvirus type 6 has also been isolated from the infections of BMT candidates (34). Therefore, screening of the patients and donors before transplantation and determining the DNA count of human herpesvirus after transplantation are essential to interpreting human viremia as a major cause of BMT failure (35).
Fungal infections are common infections in BMT recipients. The rate of fungal infections at various BMT centers has been reported to be 4 - 30% in previous studies (36). In the studies conducted in Asian countries such as India and Israel, a higher prevalence rate (19%) has also been reported (37). The emergence ratio of invasive aspergillosis has also been estimated at 7.3 - 27% in various studies (38). In a study conducted in Shiraz (Iran) in 2010, 993 blood samples were collected from BMT recipients, and 94 cases of fungal infections with Aspergillus species were reported (20). In the study by Badiee and Alborzi the prevalence of invasive aspergillosis was reported to be 15.8%, and the infection was observed to be concurrent with CMV and Aspergillus infections in five patients (20).
Hashemi and Shouhani also isolated two cases of Aspergillus flavus and one case of Trichoderma in three cases of rinsing the nasal sinuses from among 50 clinical specimens of BMT candidates suspected of fungal infections (23). In the study by Ghiasian and Kord-Bachae, 32 transplant recipients were investigated, and 15 cases of fungal infections were identified. The most frequently isolated fungal agents in the previous studies in this regard include Candida in 10 cases (66.7%), Geotrichum candidum and Cryptococcus albidus each in one case (6.7%) (22). In a study conducted in China in 2015, invasive fungal infections were evaluated in allogeneic BMT recipients, and the highest frequency of fungal agents belonged to Candida species (54.35%), followed by yeasts. Among yeasts, the highest frequency was attributed to Aspergillus (74.19%) in the mentioned study (39).
4.1. Conclusions
Although no comprehensive studies have investigated the prevalence of various infections in BMT recipients in Iran, a review of the literature in this regard indicated that among bacterial isolates, gram-positive bacteria were the most prevalent cause of infection in these patients. Among viral infectious agents in BMT recipients, CMV had the highest frequency. Among fungi, Candida and Aspergillus were the most frequent causes of fungal infections in BMT recipients, which is similar to the foreign studies in this regard. Therefore, it could be concluded that microbial infections are still a major concern in the BMT process. Due to the incidence of various disorders in the transplantation process and the postoperative phase and the risk of BMT failure, neutropenic patients and the recipients of immunosuppressants must be carefully monitored to identify the signs and symptoms of microbial infections using appropriate laboratory methods. As a result, the survival rate of these patients would increase, and treatment costs and the length of hospital stay would decrease.
References
-
1.
Lennard AL, Jackson GH. Stem cell transplantation. West J Med. 2001;175(1):42-6. [PubMed Central ID: PMC1071465]. https://doi.org/10.1136/ewjm.175.1.42.
-
2.
Lennard A, Storey N, Dickinson AM, Irving JA, Rowe D, Conn JS, et al. Collection of Philadelphia-negative peripheral blood progenitor cells in unselected patients with chronic granulocytic leukaemia. Northern Regional Haematology Group. Leukemia. 1998;12(5):746-52. [PubMed ID: 9593273]. https://doi.org/10.1038/sj.leu.2400993.
-
3.
de la Morena MT, Gatti RA. A history of bone marrow transplantation. Hematol Oncol Clin North Am. 2011;25(1):1-15. [PubMed ID: 21236386]. https://doi.org/10.1016/j.hoc.2010.11.001.
-
4.
Buckley RH, Schiff SE, Schiff RI, Markert L, Williams LW, Roberts JL, et al. Hematopoietic stem-cell transplantation for the treatment of severe combined immunodeficiency. N Engl J Med. 1999;340(7):508-16. [PubMed ID: 10021471]. https://doi.org/10.1056/NEJM199902183400703.
-
5.
Stranges E, Russo A, Friedman B. Php43 Procedures with the Most Rapidly Increasing Hospital Costs, 2004-2007. Value Health. 2010;13(3). https://doi.org/10.1016/s1098-3015(10)72424-4.
-
6.
Lee SJ, Anasetti C, Kuntz KM, Patten J, Antin JH, Weeks JC. The costs and cost-effectiveness of unrelated donor bone marrow transplantation for chronic phase chronic myelogenous leukemia. Blood. 1998;92(11):4047-52. [PubMed ID: 9834208].
-
7.
Rizzo JD, Vogelsang GB, Krumm S, Frink B, Mock V, Bass EB. Outpatient-based bone marrow transplantation for hematologic malignancies: cost saving or cost shifting? J Clin Oncol. 1999;17(9):2811-8. [PubMed ID: 10561357]. https://doi.org/10.1200/JCO.1999.17.9.2811.
-
8.
Lee SJ, Astigarraga CC, Eapen M, Artz AS, Davies SM, Champlin R, et al. Variation in supportive care practices in hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2008;14(11):1231-8. [PubMed ID: 18940677]. [PubMed Central ID: PMC3304538]. https://doi.org/10.1016/j.bbmt.2008.08.008.
-
9.
Duran-Struuck R, Dysko RC. Principles of bone marrow transplantation (BMT): providing optimal veterinary and husbandry care to irradiated mice in BMT studies. J Am Assoc Lab Anim Sci. 2009;48(1):11-22. [PubMed ID: 19245745]. [PubMed Central ID: PMC2694700].
-
10.
Cooke KR, Hill GR, Crawford JM, Bungard D, Brinson YS, Delmonte JJ, et al. Tumor necrosis factor- alpha production to lipopolysaccharide stimulation by donor cells predicts the severity of experimental acute graft-versus-host disease. J Clin Invest. 1998;102(10):1882-91. [PubMed ID: 9819375]. [PubMed Central ID: PMC509139]. https://doi.org/10.1172/JCI4285.
-
11.
Takatsuka H, Takemoto Y, Yamada S, Wada H, Tamura S, Fujimori Y, et al. Complications after bone marrow transplantation are manifestations of systemic inflammatory response syndrome. Bone Marrow Transplant. 2000;26(4):419-26. [PubMed ID: 10982289]. https://doi.org/10.1038/sj.bmt.1702517.
-
12.
Bhatia S, Francisco L, Carter A, Sun CL, Baker KS, Gurney JG, et al. Late mortality after allogeneic hematopoietic cell transplantation and functional status of long-term survivors: report from the Bone Marrow Transplant Survivor Study. Blood. 2007;110(10):3784-92. [PubMed ID: 17671231]. [PubMed Central ID: PMC2077324]. https://doi.org/10.1182/blood-2007-03-082933.
-
13.
Hows J, Bradley B. Transplantation for patients without HLA identical siblings. In: Brenner MK, Hoffbrand AV, editors. Recent advances in haematology. 8. London, UK: Churchill Livingstone; 1996. p. 191-212.
-
14.
Barnes RA, Stallard N. Severe infections after bone marrow transplantation. Curr Opin Crit Care. 2001;7(5):362-6. [PubMed ID: 11805534]. https://doi.org/10.1097/00075198-200110000-00008.
-
15.
Marr KA. Delayed opportunistic infections in hematopoietic stem cell transplantation patients: a surmountable challenge. Hematology Am Soc Hematol Educ Program. 2012;2012:265-70. [PubMed ID: 23233590]. [PubMed Central ID: PMC4696052]. https://doi.org/10.1182/asheducation-2012.1.265.
-
16.
Yazdani M, Shirani M, Baradaran S. [Tuberculosis in renal transplant recipients in Isfahan University of Medical Sciences]. J Maz Univ Med Sci. 2006;16(51):11-5. Persian.
-
17.
Saderi H, Owlia P, Moghadam K, Bahar B, Faghih Zadeh S. [Comparison of adenovirus viruria in bone marrow transplant patients before and after transplantation]. J Maz Univ Med Sci. 2005;15(48):51-7. Persian.
-
18.
Shirazi MH, Ranjbar R, Ghasemi A, Paktarigh S, Sadeghifard N, Pourmand MR. A survey of bacterial infections in bone marrow transplant recipients. Iran J Public Health. 2007;36(3):77-81.
-
19.
Bagheri K. DNA viral infections and transient bone marrow failure in southern Iran. Afr J Microbiol Res. 2012;6(36). https://doi.org/10.5897/ajmr12.725.
-
20.
Badiee P, Alborzi A. Detection of Aspergillus species in bone marrow transplant patients. J Infect Dev Ctries. 2010;4(8):511-6. [PubMed ID: 20818103]. https://doi.org/10.3855/jidc.807.
-
21.
Kokhaei P, Kokhaei L, Sadighimoghadam B, Mossayebi G. [BK virus (BKV) quantifcation in urine samples of bone marrow transplanted patients is helpful for diagnosis of hemorrhagiccystitis]. Koomesh. 2008;9(2 (26)):155-61. Persian.
-
22.
Ghiasian SA, Kord-Bachae P. [Opportunistic Fungal Infections in Haematologic Diseases and Bone Marrow Transplantation (BMT) Recipients]. Avicenna J Clin Med. 1996;3(2). Persian.
-
23.
Hashemi SJ, Shouhani M. [The comparative study of saprophytic fungi in air canal, air, hospital instruments and clinical samples from patients with bone marrow transplantation shariati hospital-2002]. Tehran Univ Med J. 2004;62(3):175-9. Persian.
-
24.
Khandan Z, Rahimi F, Akhyari H, San Ahmadi Y, Taherinia R. Frequency of CMV antigens in peripheral blood leukocytes in patients with bone marrow transplantation. J Paramed Sci. 2003;1:189-96.
-
25.
Safayi SR, Shahi F, Ghalamkari M, Mirzania M, Khatuni M, Hirmandi Niasar F. A Survey of Infection in Allogenic Hematopoietic Stem Cell Transplantation in Patients with Acute Myeloid Leukemia. Int J Organ Transplant Med. 2018;9(3):112-6. [PubMed ID: 30487958]. [PubMed Central ID: PMC6252176].
-
26.
Tousi M, Norouzy A, Vaezi M, Ghasemi A, Ghavamzade A. Microbial Evaluation of Neutropenic Diet in Bone Marrow Transplant Department in Iran. J Nutrition Fasting & Health. 2018;6(2):99-103.
-
27.
Mohammadi B, Yaghobi R, Dehghani M, Behzad Behbahani A. The molecular prevalence of viral infections in transplant candidates with bone marrow suppression, shiraz, southern iran, 2010. Int J Organ Transplant Med. 2013;4(2):87-94. [PubMed ID: 25013658]. [PubMed Central ID: PMC4089309].
-
28.
Tomas JF, Hernandez LM, Penarrubia MJ, Figuera A, Jimenez ML, Camara R, et al. [Early bacterial infections in 103 patients treated with bone marrow transplantation]. Sangre (Barc). 1994;39(3):191-6. Spanish. [PubMed ID: 7940049].
-
29.
Wingard JR. Viral infections in leukemia and bone marrow transplant patients. Leuk Lymphoma. 1993;11 Suppl 2:115-25. [PubMed ID: 8124223]. https://doi.org/10.3109/10428199309064271.
-
30.
Kawasaki H, Takayama J, Ohira M. Herpes zoster infection after bone marrow transplantation in children. J Pediatr. 1996;128(3):353-6. [PubMed ID: 8774503]. https://doi.org/10.1016/s0022-3476(96)70280-9.
-
31.
Kondo K, Kaneshima H, Mocarski ES. Human cytomegalovirus latent infection of granulocyte-macrophage progenitors. Proc Natl Acad Sci U S A. 1994;91(25):11879-83. [PubMed ID: 7991550]. [PubMed Central ID: PMC45339]. https://doi.org/10.1073/pnas.91.25.11879.
-
32.
Chen J. Animal models for acquired bone marrow failure syndromes. Clin Med Res. 2005;3(2):102-8. [PubMed ID: 16012128]. [PubMed Central ID: PMC1183440]. https://doi.org/10.3121/cmr.3.2.102.
-
33.
Bonon SH, Rossi CL, de Souza CA, Vigorito AC, Costa SC. Comparison of serology, antigenemia assay and the polymerase chain reaction for monitoring active cytomegalovirus infections in hematopoietic stem cell transplantation patients. Rev Inst Med Trop Sao Paulo. 2006;48(5):275-8. [PubMed ID: 17086316]. https://doi.org/10.1590/s0036-46652006000500007.
-
34.
Kurzrock R. Thrombopoietic factors in chronic bone marrow failure states: the platelet problem revisited. Clin Cancer Res. 2005;11(4):1361-7. [PubMed ID: 15746033]. https://doi.org/10.1158/1078-0432.CCR-04-1094.
-
35.
Lagadinou ED, Marangos M, Liga M, Panos G, Tzouvara E, Dimitroulia E, et al. Human herpesvirus 6-related pure red cell aplasia, secondary graft failure, and clinical severe immune suppression after allogeneic hematopoietic cell transplantation successfully treated with foscarnet. Transpl Infect Dis. 2010;12(5):437-40. [PubMed ID: 20561301]. https://doi.org/10.1111/j.1399-3062.2010.00515.x.
-
36.
Ninin E, Milpied N, Moreau P, Andre-Richet B, Morineau N, Mahe B, et al. Longitudinal study of bacterial, viral, and fungal infections in adult recipients of bone marrow transplants. Clin Infect Dis. 2001;33(1):41-7. [PubMed ID: 11389493]. https://doi.org/10.1086/320871.
-
37.
George B, Mathews V, Srivastava A, Chandy M. Infections among allogeneic bone marrow transplant recipients in India. Bone Marrow Transplant. 2004;33(3):311-5. [PubMed ID: 14647246]. https://doi.org/10.1038/sj.bmt.1704347.
-
38.
Marr KA, Carter RA, Boeckh M, Martin P, Corey L. Invasive aspergillosis in allogeneic stem cell transplant recipients: changes in epidemiology and risk factors. Blood. 2002;100(13):4358-66. [PubMed ID: 12393425]. https://doi.org/10.1182/blood-2002-05-1496.
-
39.
Shi JM, Pei XY, Luo Y, Tan YM, Tie RX, He JS, et al. Invasive fungal infection in allogeneic hematopoietic stem cell transplant recipients: single center experiences of 12 years. J Zhejiang Univ Sci B. 2015;16(9):796-804. [PubMed ID: 26365122]. [PubMed Central ID: PMC4569688]. https://doi.org/10.1631/jzus.B1500005.