Abstract
Background:
Staphylococcus aureus and coagulase-negative staphylococci are highly antibiotic-resistant pathogens and exhibit multiple virulence factors that threaten public health. However, few reports have evaluated the resistance and virulence of staphylococci in community settings.Objectives:
This study surveyed the epidemiology, antimicrobial resistance, and virulence genes of staphylococci in the densely-populated university town of Kunming in Southwestern China.Methods:
Various strains were isolated from hand-touching surfaces in Chenggong University Town, China, and identified via 16S rRNA sequencing. Staphylococci were isolated, and drug resistance was determined via antibiotic sensitivity testing. The mecA gene was verified and typed. Twelve virulence-associated genes and several disinfectant-resistance genes were determined.Results:
One hundred sixty-three strains were collected from hand-touching surfaces, and more than a quarter of the isolates (46/163, 28.22%) were identified as staphylococci. Among the staphylococcal strains, 19 (41.30%) showed multidrug resistance, with resistance rates of more than 40% to benzyl-penicillin, erythromycin, oxacillin, and tetracycline. Twenty isolates (43.48%) carried mecA, and most staphylococcal cassette chromosome mecs (SCCmecs) were nontypeable. Twenty-eight strains (60.87%) carried at least one virulence gene; hemolysin genes were the most prevalent, and 22 strains (47.83%) carried qac genes. Staphylococcus aureus strain ST59 carried multiple resistance and virulence genes.Conclusions:
Resistant staphylococci can easily adhere to hand-touching surfaces. More attention should be given to the appearance of resistance and virulence gene-harboring staphylococci in community settings. The findings herein suggest that effective disinfectant strategies and feasible surveillance measures are urgently needed.Keywords
Hand-Touching Surface Staphylococci Virulence Resistance mecA
1. Background
Staphylococci are common Gram-positive bacteria that inhabit community and hospital settings. Among all staphylococci, Staphylococcus aureus is the most notorious coagulase-positive Staphylococcus sp. and causes severe clinical infections and food poisoning (1, 2). In addition, the heterogeneous group of coagulase-negative staphylococci (CoNS) has become increasingly prevalent, and CoNS are listed as major nosocomial opportunistic pathogens (3). Antimicrobial resistance is a global issue. Due to increasing resistance, community-isolated staphylococci may become a severe threat to public health, with delayed treatment opportunities and higher costs. Oxacillin and cefoxitin are commonly used to treat staphylococcal infections. Expression of the mecA gene, which encodes the low-affinity penicillin-binding protein, PBP-2a (4), was reported to be the main reason that staphylococci show resistance to oxacillin and cefoxitin. Thus, mecA gene expression inhibits treatment of clinical infections (5). Typing the staphylococcal cassette chromosome mec (SCCmec), the mobile genetic element carrying mecA, can provide a detailed genetic linkage of mecA (6).
Reports on staphylococcal virulence have mainly concentrated on staphylococci in food, but few reports have concentrated on staphylococci in community settings. Staphylococci can produce multiple toxins, including staphylococcal enterotoxin (SE), hemolysin (HL), and Panton-Valentine leukocidin (PVL). SEs damage the host immune system by targeting the innate and adaptive responses (7), and HL and PVL can damage cell membranes, causing severe diseases (8, 9). Increasing numbers of staphylococcal surveillance systems are being reported in community settings, such as metro systems (10), hotels (11), washrooms (12), and other non-healthcare settings (13), but few staphylococcal surveillance systems have been reported on densely populated campuses. Chenggong University Town in Kunming city in Southwestern China was built in 2010.
Ten universities and colleges have entered this university town and accommodate more than one hundred thousand students. The high population density provides ideal conditions for staphylococcal transmission via hand-touching surfaces. If the staphylococci on these surfaces contain virulence and drug-resistance genes, these bacteria could threaten community public health. Therefore, understanding the molecular characteristics, microbiology and epidemiology of the staphylococci isolated from hand-touching surfaces in this university town is crucial.
2. Objectives
In this study, we surveyed the epidemiology, antimicrobial resistance and virulence of staphylococci isolated from hand-touching surfaces in Chenggong University Town in Southwestern China.
3. Methods
3.1. Strain Collection and Identification
Samples were collected from hand-touching surfaces from six universities in Chenggong University Town from March 2016 to May 2016. The high-frequency hand-touching surfaces were predetermined to include stair rails, elevator buttons, campus card transfer machines, automated teller machines, door handles, classroom desks, library entrance machines, and refrigerator handles in convenience stores. The swab sampling method was followed as previously described (10). Each swab was placed in a sterile tube with 5 mL saline to dilute the bacterial concentration, then transferred to Luria-Bertani agar plates and incubated for 18 - 24 hours. Single clones were selected and incubated with Luria-Bertani broth for another 24 hours. Genomic DNA was obtained using a TIANamp Bacterial DNA Kit (Tiangen Biotech Co., Ltd., Beijing, China) per the manufacturer’s instructions. Genealogical classifications of the 16S rRNA were determined for each strain via PCR amplification and 27F/1492R primer sequencing (27F: 5’-AGAGTTTGATCCTGGCTCAG-3’, 1492R: 5’-GGTTACCTTGTTACGACTT-3’). The sequences were blasted in the NCBI database for species identification. Nonduplicated colonies identified as Staphylococcus were selected.
3.2. Antimicrobial Susceptibility Testing
Antimicrobial susceptibility tests were performed on all collected staphylococci using the VITEK-2 compact (bioMérieux, Marcy l’Etoile, France). The antimicrobial tests included the primary test, which reported resistance to erythromycin, clindamycin, oxacillin, benzylpenicillin, and trimethoprim-sulfamethoxazole, and tests for resistance to other routinely reported antimicrobials, including gentamicin, ciprofloxacin, levofloxacin, moxifloxacin, quinupristin-dalfopristin, linezolid, vancomycin, tetracycline, tetracycline, nitrofurantoin, and rifampicin. In addition, the VITEK-2 compact provided the cefoxitin screening test for detecting methicillin-resistant strains mediated by mecA. The antimicrobial susceptibility testing interpretive standard followed the guidelines of the Clinical and Laboratory Standard Institute, M100S 27th.
3.3. Resistance Gene Detection and mecA Typing
The mecA gene was detected and identified in all staphylococcal strains via polymerase chain reaction (PCR) assays. For the mecA-harboring strains, multiplex PCR was used to type the SCCmec, and mecA was classified as types I - V or nontypeable as previous report (6). Table 1 lists the primers used for the PCR. We further surveyed the multilocus sequence typing of S. aureus by amplifying and sequencing seven housekeeping genes (https://pubmlst.org/saureus/). The anti-disinfectant-related gene, qac, was also detected.
The Primer Used for Detection of Resistance and Virulence Genes
| Primers | Primer Sequences(5’-3’) | Base Pairs | Reference |
|---|---|---|---|
| mecA gene | |||
| mecA-F | GTTGTAGTTGTCGGGTTT | 437 | (14) |
| mecA-R | CCACATTGTTTCGGTCTA | ||
| Anti-disinfectant | |||
| qac-F | CAGTTTGTAATTGGAGGC | 429 | (14) |
| qac-R | TTTCTTTGATAGCTGCTTG | ||
| Exotoxins | |||
| seA-F | TTGGAAACGGTTAAAACGAA | 120 | (15) |
| seA-R | GAACCTTCCCATCAAAAACA | ||
| seB-F | TCGCATCAAACTGACAAACG | 478 | (15) |
| seB-R | GCGGTACTCTATAAGTGCC | ||
| seE-F | AGGTTTTTTCACAGGTCATCC | 209 | (15) |
| seE-R | CTTTTTTTTCTTCGGTCAATC | ||
| seG-F | AAGTAGACATTTTTGGCGTTCC | 287 | (15) |
| seG-R | AGAACCATCAAACTCGTATAGC | ||
| seH-F | GTCTATATGGAGGTACAACACT | 213 | (15) |
| seH-R | GACCTTTACTTATTTCGCTGTC | ||
| seI-F | GGTGATATTGGTGTAGGTAAC | 454 | (15) |
| seI-R | ATCCATATTCTTTGCCTTTACCAG | ||
| seU-F | AAACATTAAAGCCCAAGAG | 241 | This study |
| seU-R | ACCGCCATACATACACG | ||
| Hemolysin | |||
| hlA-F | ATGGTGAATCAAAATTGGGG | 205 | (16) |
| hlA-R | GTTGTTTGGATGCTTTTC | ||
| hlB-F | GCCAAAGCCGAATCTAAG | 241 | (16) |
| hlB-R | CGAGTACAGGTGTTTGGT | ||
| hlG-F | CTCTTGCCAATCCGTTATTA | 253 | (16) |
| hlG-R | GCTTTAACATGATTAGTTTT | ||
| hlD-F | TGACAGTGAGGAGAGTGGTGT | 254 | This study |
| hlD-R | GTGCACCATGTGCATGTCTT | ||
| Panton-Valentine leukocidin | |||
| pvl-F | ACACACTATGGCAATAGTTA TTT | 176 | (17) |
| pvl-R | AAAGCAATGCAATTGATGTA | ||
| Coagulase | |||
| cal-F | GAGATACAGACAATCCACATAA | 601 | (18) |
| cal-R | CTACCTTCAAGACCTTCTAAAA |
3.4. Virulence Gene Checking
The CoNS isolates were confirmed by verifying the coagulase gene, cal. Common toxin-producing genes were also detected in all staphylococcal strains, including staphylococcal enterotoxin genes (seA, seB, seE, seG, seH, seI, and seU), staphylococcal hemolysin genes (HLa, HLb, HLd, and HLg), and the Panton-Valentine leukocidin gene (pvl). Table 1 lists the primers used to verify the virulence genes.
4. Results
4.1. Isolation of Staphylococci
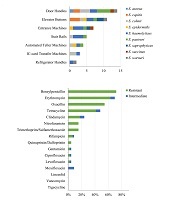
From March 2016 to May 2016, 163 strains belonging to diverse genera, including Bacillus, Micrococci, and Kocuria, were collected and identified. Among them, 46 strains (28.22%) were identified as staphylococci, and nine distinct species were detected according to the genealogical classification results of the 16S rRNA sequencing: S. aureus (1/46, 2.17%), S. capitis (5/46, 10.87%), S. cohnii (8/46, 17.39%), S. epidermidis (3/46, 6.52%), S. haemolyticus (13/46, 28.26%), S. pasteuri (8/46,17.39%), S. saprophyticus (3/46, 6.52%), S. succinus (2/46, 4.35%), and S. warneri (3/46, 6.52%) (Figure 1). All staphylococcal species were CoNS, except S. aureus. Figure 2A shows the predetermined seven locations from which the staphylococci were obtained.
Proportion of all isolates and staphylococci species

A, the locational distribution and B, the drug-resistance rate

4.2. Antimicrobial Susceptibility Testing
Sixteen antibiotics were used to test the antimicrobial susceptibility of the 46 collected staphylococcal strains. The 46 staphylococcal strains showed high resistance rates of over 40% to four commonly used antimicrobials (benzyl-penicillin, erythromycin, oxacillin and tetracycline) and resistance rates of less than 20% to clindamycin, nitrofurantoin, trimethoprim/sulfamethoxazole, rifampicin, quinupristin-dalfopristin, gentamicin, ciprofloxacin and levofloxacin (Figure 2B). None of the strains showed resistance to linezolid, vancomycin or tigecycline. Strains that displayed resistance to at least three antimicrobial classes were categorized as multidrug-resistant (MDR). Of the 46 staphylococci, 41.30% (19/46) were MDR. The S. aureus strain was identified as MDR and showed resistance to benzyl-penicillin, oxacillin, erythromycin, clindamycin, and tetracycline.
4.3. Molecular Epidemiological Characteristics of Resistance Genes
Table 2 shows the mecA and qac detection rates. Of the 25 oxacillin-resistant staphylococci, 20 isolates carried mecA and tested positive for cefoxitin resistance, but the other 5 isolated oxacillin-resistant staphylococci did not. Among the mecA carriers, 60.00% (12/20) were categorized as MDR. SCCmec typing showed that two mecA carriers were SCCmec type III, one was SCCmec type IVc, and the remaining seventeen were nontypeable (Figure 3). The nontypeable SCCmec staphylococci showed a wide species distribution and varying minimum inhibitory concentrations (MICs; 0.125 to ≥ 4 μg/mL) to beta-lactams (Table 3). In addition, 22 strains carried the anti-disinfectant-related gene, qac. The oxacillin-resistant S. aureus strain carrying the qac and mecA (type IVc) genes was considered methicillin-resistant S. aureus (MRSA), and further multilocus sequence typing showed that this strain was ST59.
The Carry Rates of Resistance and Virulence Genes
| Class | Gene | Total | Percent |
|---|---|---|---|
| Resistance | mecA | 20 | 43.48 |
| qac | 23 | 50.00 | |
| Virulence | seA | 5 | 10.87 |
| seB | 3 | 6.52 | |
| seE | 0 | 0.00 | |
| seG | 0 | 0.00 | |
| seI | 3 | 6.52 | |
| seH | 1 | 2.17 | |
| seU | 0 | 0.00 | |
| hlA | 12 | 26.09 | |
| hlB | 6 | 13.04 | |
| hlD | 8 | 17.39 | |
| hlG | 16 | 34.78 | |
| pvl | 3 | 6.52 | |
| cal | 1 | 2.17 |
SCCmec Types and MIC Ranges Among mecA Carrying Staphylococci
| Species | SCCmec | No. of Isolates | MIC Range (μg/mL) | |
|---|---|---|---|---|
| Benzylpenicillin | Oxacillin | |||
| S. aureus | IVc | 1 | ≥ 0.5 | 0.5 |
| S. capitis | Nontypeable | 3 | ≥ 0.5 | 0.5 ~ ≥ 4 |
| S. cohnii | Nontypeable | 4 | 0.125 ~ ≥ 0.5 | 0.5 ~ 1 |
| S. epidermidis | III | 1 | ≥ 0.5 | 0.5 |
| S. haemolyticus | Nontypeable | 3 | 0.25 ~ ≥ 0.5 | 0.25 ~ 0.5 |
| S. haemolyticus | III | 1 | ≥ 0.5 | 0.5 |
| S. pasteuri | Nontypeable | 3 | ≥ 0.5 | 0.5 ~ ≥ 4 |
| S. saprophyticus | Nontypeable | 2 | 0.125 ~ 0.25 | 0.5 ~ 2 |
| S. succinus | Nontypeable | 1 | 0.25 | 1 |
| S. warneri | Nontypeable | 1 | ≥ 0.5 | ≥ 4 |
PCR gel electrophoresis of mecA typing for the mecA harboring strains

4.4. Molecular Epidemiology of the Virulence Genes
Virulence gene detection revealed that 28 strains (28/46) harbored at least one virulence gene. Seven strains carried only staphylococcal enterotoxin genes, 15 strains carried only hemolysin genes, and one strain carried only pvl. Moreover, three strains carried both se and hl but not pvl, and one strain carried pvl and se but not hl. The S. aureus strain carried seB, hlA, hlB, hlD, hlG and pvl in addition to cal, but no CoNS strains carried cal. Table 2 shows that hlG (16/46) was the most frequently detected virulence gene, followed by hlA (12/46), hlD (8/46), hlB (6/46), seA (5/46), seB (3/46), seI (3/46), pvl (3/46) and she (1/46), while seE, seG and seU were undetected.
5. Discussion
Staphylococci are common widespread Gram-positive bacteria that are reported to be a significant threat for nosocomial infections because of their drug resistance and an important food-safety threat because of their virulence. In the current study, we surveyed the prevalence of staphylococci isolated from hand-touching surfaces in the new Chennggong University Town and further characterized the epidemiology of staphylococcal resistance and virulence.
The selected sampling sites were in public locations and could be touched by random people. We cultivated the samples using Luria-Bertani (L-B) agar plates, which can be used to incubate and culture most bacteria, including common opportunistic pathogens. The samples were diluted and transferred to L-B agar plates, which enabled determining the staphylococcal proportions in the L-B-culturable environmental samples. We acquired more than twenty bacterial species that were common in the environment, and, other than the staphylococci, most isolates showed no or few harmful effects to humans. Forty-six staphylococcal isolates, followed by the most easily-isolated Bacillus spp. (57/163, 34.97%), accounted for more than a quarter of all isolates (46/163, 28.22%), implying that staphylococci commonly adhere to hand-touching surfaces around the university.
Door handles, elevator buttons and entrance machines were the top 3 locations from which staphylococci were collected (Figure 2A). These locations all had metal surfaces, suggesting that staphylococci adhere more easily to metal surfaces than do other bacterial species. In addition, few S. aureus isolates were obtained in this study; however, as previous studies reported, S. aureus is more easily isolated from food (isolated rates of 17.7%) (19), and food (especially meat) may provide better growth conditions for S. aureus (20, 21). In addition, insects such as cockroaches may serve as media for bacterial transmission (22). However, in this study, the hand-touching surfaces lacked sufficient nutritional conditions, which might be more suitable for community-related staphylococcal adherence.
Drug-resistant bacteria are receiving increasing attention in clinical infection control because of their adverse effects, including delayed healing and higher treatment costs (23). In this study, 33 isolates (71.74%) were resistant to benzyl-penicillin, 29 (63.04%) to erythromycin, 25 (54.35%) to oxacillin, and 19 (41.30%) to tetracycline. These were the four highest resistance rates, and all were over 40%. A previous study (14) also found that community-acquired Staphylococcus had higher resistance rates to oxacillin (27.90%), G-penicillin (26.74%), and erythromycin (22.09%) than to other antibiotics, but these rates were lower than those found in the present study. Additionally, 19 strains (41.30%) showed characteristics of multidrug resistance, which was similar to the results of a previous report (49.6%) (24), but was higher than that found for food-source strains (20.73%) (25).
The high resistance rates to commonly used antibiotics and the existence of MDR strains suggest an increasing threat to public health due to community-acquired Staphylococcus in this university town. Nevertheless, the resistance rates of these community-acquired strains to commonly used drugs were lower than those of clinical strains (26). Furthermore, no strains showed resistance to linezolid, vancomycin or tigecycline, which are considered the final effective agents for controlling staphylococcal infections. Therefore, although resistance to community-acquired staphylococci threatens public health, strategies against staphylococcal infections are available.
The mecA gene is important for conferring staphylococcal resistance to beta-lactam antimicrobials. Eleven SCCmec types have been reported for S. aureus, but some reports have shown that CoNS harbor a greater variety of SCCmec genes, enabling various mecA genes to be exchanged between S. aureus and CoNS strains (27). Among the mecA-harboring CoNS strains, except for one SCCmec type III strain and one SCCmec type IVc strain, 17 mecA genes could not be classified via current approaches, suggesting that environmental staphylococci may have greater SCCmec diversity than that of clinical isolates. In addition to carrying mecA, multiple other mechanisms can lead to oxacillin resistance, and 5 non-mecA-harboring strains have shown oxacillin resistance due to other mechanisms, such as biofilm formation, which has been previously described (28).
More than half the staphylococcal strains (28/46) carried at least one virulence gene, and hemolysin genes were the most prevalent (20/28), suggesting that these community-acquired staphylococci can easily cause infections. Of these strains, 26.09% (12/46) carried staphylococcal enterotoxin genes, whereas a larger number of food-source strains (20/24) carried these genes (15). Nearly half the toxin-producing staphylococcal strains (13/28) harbored mecA, suggesting that these antibiotic-resistant strains, together with toxic strains, are potential burdens to public health, and the hand-touching surfaces from which they were isolated should be sterilized. However, the qac gene was prevalent in more than half (7/13) of the mecA and virulence gene-harboring strains, and another 16 strains carried the qac gene; thus, various disinfection methods should be considered.
In this study, CoNS were the most easily isolated pathogenic bacteria. Other than the CoNS, pathogenic bacteria were detected at low rates in this university town. Only one Pseudomonas aeruginosa strain and one S. aureus strain were detected among the 163 collected strains, and these two species are considered opportunistic pathogens commonly detected in clinical settings. Although only one S. aureus strain was isolated, further analysis revealed that this strain was a threat to public health. Multilocus sequence typing showed that this classroom-isolated strain was ST59, which is mostly isolated from clinical settings but less often from environmental settings as described in the database, and it once caused severe pneumonia in China (29).
In the current study, strain ST59 was identified as MRSA via antimicrobial susceptibility testing, and this strain was carrying type IVc mecA. As previously described (30), ST59 can harbor multiple SCCmec types such as IV, V, VII and VIII. CoNS strains isolated from the same place may carry many SCCmec types, which may enable exchanging mecA with S. aureus ST59. Furthermore, the seB, hlA, hlB, hlD, hlG and pvl virulence genes were detected, which conferred infection and food-poisoning abilities to this MRSA strain. Multiple virulence factors and multidrug resistance, together with harboring the qac gene, enable ST59 to easily invade the human body and be difficult to disinfect.
5.1. Conclusions
In conclusion, this study demonstrated that multidrug-resistant Staphylococcus is commonly scattered across hand-touching surfaces in Chenggong University Town. Virulence, antibiotic resistance and disinfectant resistance are prevalent among these staphylococci and can potentially threaten food safety, infection treatments and public health. To reduce the risk of infection by staphylococci, which adhere to and are transmitted on hand-touching surfaces, more effective disinfectant strategies are urgently needed, and feasible surveillance measures should be adopted.
References
-
1.
Denayer S, Delbrassinne L, Nia Y, Botteldoorn N. Food-borne outbreak investigation and molecular typing: High diversity of Staphylococcus aureus strains and importance of toxin detection. Toxins (Basel). 2017;9(12). [PubMed ID: 29261162]. [PubMed Central ID: PMC5744127]. https://doi.org/10.3390/toxins9120407.
-
2.
Thomer L, Schneewind O, Missiakas D. Pathogenesis of Staphylococcus aureus bloodstream infections. Annu Rev Pathol. 2016;11:343-64. [PubMed ID: 26925499]. [PubMed Central ID: PMC5068359]. https://doi.org/10.1146/annurev-pathol-012615-044351.
-
3.
Khan MM, Faiz A, Ashshi AM. Clinically significant coagulase negative Staphylococci and their antibiotic resistance pattern in a tertiary care hospital. J Pak Med Assoc. 2014;64(10):1171-4. [PubMed ID: 25823159].
-
4.
Zong Z, Peng C, Lu X. Diversity of SCCmec elements in methicillin-resistant coagulase-negative Staphylococci clinical isolates. PLoS One. 2011;6(5). e20191. [PubMed ID: 21637845]. [PubMed Central ID: PMC3102682]. https://doi.org/10.1371/journal.pone.0020191.
-
5.
Rolo J, Worning P, Boye Nielsen J, Sobral R, Bowden R, Bouchami O, et al. Evidence for the evolutionary steps leading to mecA-mediated beta-lactam resistance in Staphylococci. PLoS Genet. 2017;13(4). e1006674. [PubMed ID: 28394942]. [PubMed Central ID: PMC5402963]. https://doi.org/10.1371/journal.pgen.1006674.
-
6.
Zhang K, McClure JA, Elsayed S, Louie T, Conly JM. Novel multiplex PCR assay for characterization and concomitant subtyping of staphylococcal cassette chromosome mec types I to V in methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 2005;43(10):5026-33. [PubMed ID: 16207957]. [PubMed Central ID: PMC1248471]. https://doi.org/10.1128/JCM.43.10.5026-5033.2005.
-
7.
Argudin MA, Mendoza MC, Rodicio MR. Food poisoning and Staphylococcus aureus enterotoxins. Toxins (Basel). 2010;2(7):1751-73. [PubMed ID: 22069659]. [PubMed Central ID: PMC3153270]. https://doi.org/10.3390/toxins2071751.
-
8.
Vandenesch F, Lina G, Henry T. Staphylococcus aureus hemolysins, bi-component leukocidins, and cytolytic peptides: A redundant arsenal of membrane-damaging virulence factors? Front Cell Infect Microbiol. 2012;2:12. [PubMed ID: 22919604]. [PubMed Central ID: PMC3417661]. https://doi.org/10.3389/fcimb.2012.00012.
-
9.
Prevost G, Cribier B, Couppie P, Petiau P, Supersac G, Finck-Barbancon V, et al. Panton-Valentine leucocidin and gamma-hemolysin from Staphylococcus aureus ATCC 49775 are encoded by distinct genetic loci and have different biological activities. Infect Immun. 1995;63(10):4121-9. [PubMed ID: 7558328]. [PubMed Central ID: PMC173579].
-
10.
Peng Y, Ou Q, Lin D, Xu P, Li Y, Ye X, et al. Metro system in Guangzhou as a hazardous reservoir of methicillin-resistant Staphylococci: Findings from a point-prevalence molecular epidemiologic study. Sci Rep. 2015;5:16087. [PubMed ID: 26513750]. [PubMed Central ID: PMC4625601]. https://doi.org/10.1038/srep16087.
-
11.
Xu Z, Mkrtchyan HV, Cutler RR. Antibiotic resistance and mecA characterization of coagulase-negative Staphylococci isolated from three hotels in London, UK. Front Microbiol. 2015;6:947. [PubMed ID: 26441881]. [PubMed Central ID: PMC4563241]. https://doi.org/10.3389/fmicb.2015.00947.
-
12.
Mkrtchyan HV, Xu Z, Cutler RR. Diversity of SCCmec elements in Staphylococci isolated from public washrooms. BMC Microbiol. 2015;15:120. [PubMed ID: 26071078]. [PubMed Central ID: PMC4465472]. https://doi.org/10.1186/s12866-015-0451-3.
-
13.
Xu Z, Shah HN, Misra R, Chen J, Zhang W, Liu Y, et al. The prevalence, antibiotic resistance and mecA characterization of coagulase negative Staphylococci recovered from non-healthcare settings in London, UK. Antimicrob Resist Infect Control. 2018;7:73. [PubMed ID: 29946448]. [PubMed Central ID: PMC6000976]. https://doi.org/10.1186/s13756-018-0367-4.
-
14.
Zhou F, Wang Y. Characteristics of antibiotic resistance of airborne Staphylococcus isolated from metro stations. Int J Environ Res Public Health. 2013;10(6):2412-26. [PubMed ID: 23765189]. [PubMed Central ID: PMC3717744]. https://doi.org/10.3390/ijerph10062412.
-
15.
Madahi H, Rostami F, Rahimi E, Safarpoor Dehkordi F. Prevalence of enterotoxigenic Staphylococcus aureus isolated from chicken nugget in Iran. Jundishapur J Microbiol. 2014;7(8). e10237. [PubMed ID: 25485044]. [PubMed Central ID: PMC4255206]. https://doi.org/10.5812/jjm.10237.
-
16.
Zhang H, Zheng Y, Gao H, Xu P, Wang M, Li A, et al. Identification and characterization of Staphylococcus aureus strains with an incomplete hemolytic phenotype. Front Cell Infect Microbiol. 2016;6:146. [PubMed ID: 27917374]. [PubMed Central ID: PMC5114236]. https://doi.org/10.3389/fcimb.2016.00146.
-
17.
McDonald RR, Antonishyn NA, Hansen T, Snook LA, Nagle E, Mulvey MR, et al. Development of a triplex real-time PCR assay for detection of Panton-Valentine leukocidin toxin genes in clinical isolates of methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 2005;43(12):6147-9. [PubMed ID: 16333116]. [PubMed Central ID: PMC1317231]. https://doi.org/10.1128/JCM.43.12.6147-6149.2005.
-
18.
Dai Y, Chang W, Zhao C, Peng J, Xu L, Lu H, et al. Vrar binding to the promoter region of agr inhibits its function in vancomycin-intermediate Staphylococcus aureus (VISA) and heterogeneous VISA. Antimicrob Agents Chemother. 2017;61(5). [PubMed ID: 28289032]. [PubMed Central ID: PMC5404600]. https://doi.org/10.1128/AAC.02740-16.
-
19.
Hasanpour Dehkordi A, Khaji L, Sakhaei Shahreza MH, Mashak Z, Safarpoor Dehkordi F, Safaee Y, et al. One-year prevalence of antimicrobial susceptibility pattern of methicillin-resistant Staphylococcus aureus recovered from raw meat. Trop Biomed. 2017;34(2):396-404.
-
20.
Safarpoor Dehkordi F, Akhondzadeh Basti A, Gandomi H, Misaghi A, Rahimi E. Pathogenic Staphylococcus aureus in hospital food samples; prevalence and antimicrobial resistance properties. J Food Safe. 2018;38(6). e12501. https://doi.org/10.1111/jfs.12501.
-
21.
Safarpoor Dehkordi F, Gandomi H, Basti AA, Misaghi A, Rahimi E. Phenotypic and genotypic characterization of antibiotic resistance of methicillin-resistant Staphylococcus aureus isolated from hospital food. Antimicrob Resist Infect Control. 2017;6:104. [PubMed ID: 29034091]. [PubMed Central ID: PMC5628482]. https://doi.org/10.1186/s13756-017-0257-1.
-
22.
Abdolmaleki Z, Mashak Z, Safarpoor Dehkordi F. Phenotypic and genotypic characterization of antibiotic resistance in the methicillin-resistant Staphylococcus aureus strains isolated from hospital cockroaches. Antimicrob Resist Infect Control. 2019;8:54. [PubMed ID: 30911380]. [PubMed Central ID: PMC6416839]. https://doi.org/10.1186/s13756-019-0505-7.
-
23.
Rowan MP, Cancio LC, Elster EA, Burmeister DM, Rose LF, Natesan S, et al. Burn wound healing and treatment: Review and advancements. Crit Care. 2015;19:243. [PubMed ID: 26067660]. [PubMed Central ID: PMC4464872]. https://doi.org/10.1186/s13054-015-0961-2.
-
24.
Stepanovic S, Cirkovic I, Djukic S, Vukovic D, Svabic-Vlahovic M. Public transport as a reservoir of methicillin-resistant Staphylococci. Lett Appl Microbiol. 2008;47(4):339-41. [PubMed ID: 19241530]. https://doi.org/10.1111/j.1472-765x.2008.02436.x.
-
25.
Momtaz H, Safarpoor Dehkordi F, Rahimi E, Asgarifar A, Momeni M. Virulence genes and antimicrobial resistance profiles of Staphylococcus aureus isolated from chicken meat in Isfahan province, Iran. J Appl Poultr Res. 2013;22(4):913-21. https://doi.org/10.3382/japr.2012-00673.
-
26.
Deng Y, Liu J, Peters BM, Chen L, Miao J, Li B, et al. Antimicrobial resistance investigation on Staphylococcus strains in a local hospital in Guangzhou, China, 2001-2010. Microb Drug Resist. 2015;21(1):102-4. [PubMed ID: 25299244]. [PubMed Central ID: PMC4442556]. https://doi.org/10.1089/mdr.2014.0117.
-
27.
Saber H, Jasni AS, Jamaluddin T, Ibrahim R. A review of staphylococcal cassette chromosome mec (SCCmec) types in coagulase-negative Staphylococci (CoNS) species. Malays J Med Sci. 2017;24(5):7-18. [PubMed ID: 29386968]. [PubMed Central ID: PMC5772811]. https://doi.org/10.21315/mjms2017.24.5.2.
-
28.
Seng R, Kitti T, Thummeepak R, Kongthai P, Leungtongkam U, Wannalerdsakun S, et al. Biofilm formation of methicillin-resistant coagulase negative staphylococci (MR-CoNS) isolated from community and hospital environments. PLoS One. 2017;12(8). e0184172. [PubMed ID: 28859149]. [PubMed Central ID: PMC5578677]. https://doi.org/10.1371/journal.pone.0184172.
-
29.
Qu T, Feng Y, Jiang Y, Zhu P, Wei Z, Chen Y, et al. Whole genome analysis of a community-associated methicillin-resistant Staphylococcus aureus ST59 isolate from a case of human sepsis and severe pneumonia in China. PLoS One. 2014;9(2). e89235. [PubMed ID: 24586619]. [PubMed Central ID: PMC3930696]. https://doi.org/10.1371/journal.pone.0089235.
-
30.
Figueiredo AM, Ferreira FA. The multifaceted resources and microevolution of the successful human and animal pathogen methicillin-resistant Staphylococcus aureus. Mem Inst Oswaldo Cruz. 2014;109(3):265-78. [PubMed ID: 24789555]. [PubMed Central ID: PMC4131778]. https://doi.org/10.1590/0074-0276140016.