Abstract
Background:
Despite using various chemotropic drugs, the therapy strategies of cervical cancer are not satisfactory. Researchers tended to examine anti-proliferative effects of different botanical components such as crocin on several cancers.Objectives:
We investigated the anti-cancer mechanisms of crocin and epirubicin combination on cancer cervical cells to use fewer toxic components.Methods:
By MTT, Hoechst staining and flowcytometry techniques, cell proliferation inhibitory, and apoptosis induction effects of crocin and epirubisin combination on a cervical cancer cell line (OV2008) were determined. Then, alteration expression of the Bax, Bcl2, and P21 genes were also investigated through real-time polymerase chain reaction (real-time PCR).Results:
The MTT results showed that crocin and epirubicin stopped proliferation of OV2008 cells. These substances had a simple additive effect after 72 hours treatment. The Hoechst staining illustrated that apoptosis significantly increased after combination therapy. The results of the cell cycle analysis showed that the percentage of dead cells was increased compared to the control group. Also, the number of cells, introduced in all of cell cycle phases, was changed, too. Moreover, real-Time PCR results showed that epiburicin 3.75 µm/mL and crocin 2000 µm/mL led to Bax and P21 up-regulated and Bcl2 down-regulated.Conclusions:
Crocin can be used in companion with fewer dose of chemotropic drugs to get better results in cancer therapy and can be more effective on the reduction of drug resistance due to target the main regulators of apoptosis. Nonetheless, more surveys are needed to enlighten their molecular mechanisms.Keywords
1. Background
Nowadays, the prevalence of cervical cancer and its death rate are still high among women around the world and has the forth rank in incidence as well as mortality (1, 2). Although there are many strategies for declining its symptoms such as surgery, radiotherapy, chemotherapy, and their combination following tumor resection and recurrence is usually occurring due to cancer cell resistance to therapy (3). Also, damaged normal cells are another drawback of these therapies. Hence, today many studies have been accomplished to investigate the anti-cancer characteristics of natural agents such as medicinal plants (4-6).
Crocin (C44H70O28) with molecular weight of 976.96 g/mol is one of the main water-solving carotenoids of saffron stigma (7). Several investigations illustrated that crocin could be used for curing many diseases such as cancers (8), neurological disorders (9), cognitive complications (10), immune system diseases (11-13), hepatocyte damages (14), and many kinds of inflammation (15). In the last decades, many researchers investigated the anti-proliferative properties of crocin on different cancers through different pathways, such as apoptosis (7). Sun et al. stated in their survey that crocin could prohibit the growth and tumorigenicity of HL-60 cancer cells (16). Before them, in 1996 Escribano et al. compared the invasive effects of 4 compounds of saffron on Hela cells, too; crocin was the most effective one (17). Aung et al. also demonstrated that crocin had anti-proliferative effects on some cancer colorectal cell lines (18). In previous studies, anti-growth and pro-cell death effects of crocin have been reported In-vivo (19) and In-vitro (20). Likewise, changes in the Bax/Bcl-2 ratio, activation of caspases, and cell cycle arrest by an alteration in cyclin D1 and p21 mRNA levels were studied (21). Crocin can be effective in cell cycle arrest at G0/G1 phase via changing the expression of some key proteins in different cancers such as Bax and Bcl2 (22). However, its anti-cancer molecular mechanism is not yet completely known. So, crocin notably has a chemo-preventive activity because of its low toxicity and potent efficacy in cancer (23).
2. Objectives
We aimed at assessing the inhibitory effect of this carotenoid in combination with epirubicin as one of the main chemotherapy drugs, on the proliferation of cervical cancer cells for first time.
3. Methods
3.1. Crocin Preparation
According to our recent studies, Crocin was isolated and purified from Iranian saffron (21).
3.2. Cell Cultures
The OV2008 cells were kindly prepared from Prof. Tsang (University of Ottawa, Canada) as chemo-sensitive cervical cancer cells. They were cultured in RPMI1640 cell media supplemented with 10% inactivated FBS, 100 units/mL penicillin, and 100 mg/mL streptomycin (Gibco BRL, Grand Island, NY, USA). After that, the cells were treated with crocin in different concentration (0 - 4000 µm/mL) and epirubicin (0 - 6.25 µm/mL) combination (R = 3) and incubated at 37°C including the atmosphere of 5% CO2.
3.3. Cell Viability Assay
Crocin cytotoxicity effects on OV2008 were measured by MTT assay (Sigma-Aldrich; M2128-1G, USA) according to our former study (3). The absorbance was read at 540 nm by ELISA plate reader (BioTek, Epoch, USA). Furthermore, the IC50 (initial concentration of component that decreased the percentages of live cells to 50%) of these combinations was determined to analyze the cytotoxic efficiency of them by using the dose- and time-dependent curves.
3.4. Appraisement of Apoptosis with Hoechst Staining
After exposing the cells with Crocin and Epirubicin, they were harvested in 6 wells plates and stained by Hoechst 33258 (12.5 ng/mL) to study the percentages of apoptotic nuclear morphology (nuclear shrinkage, condensation, and fragmentation) (20).
To measure synergic effect of crocin and epirubicin, the synergic index (q) was calculated with below formula according to our previous studies (3, 24):
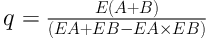
E (A + B) showed the growth suppression rate of crocin and epirubicin. EA and EB were their suppression rates alone, respectively.
0.85 < q < 1.15: Simple addictive effects
1.15 < q < 2: A degree of synergism
q > 2: Significant synergistic effect
3.5. RNA Extraction and Reverse Transcription Polymerase Chain Reaction (RT-PCR)
RNX Plus reagent (Cinnagen, Iran) was used to extract RNA, entirely. 260/280 and 260/230 absorbance ratio were achieved, using nanodrop (BioTek, Epoch, USA). The primer sequences are as follow: β-actin (forward: TGGCACCCAGCACAATGAA, reverse: CTAAGTCATAGTCCGCCTAGAAGCA), Bax (forward: GGTTGTCGCCCTTTTCTA, reverse: CGGAGGAAGTCCAATGTC), Bcl2 (forward: GATGTGATGCCTCTGCGAAG, reverse: CATGCTGATGTCTCTGGAATCT), P21 (forward: GGCAGACCAGCATGACAGATT, reverse: GCGGATTAGGGCTTCCTCTT). The cDNA was synthesized by PrimeScript RT™ reagent kit (Thermo Scientific, USA). The quantities RT-PCR was done by the SYBRGreen master mix (Parstoos, Iran) and ABI Step One TM Real-Time PCR System (Applied Biosystems, Foster City, CA) (3, 24).
3.6. Propidium Iodide (PI) Staining Assay
PI staining kit (IQ product, Netherlands) was used for apoptosis detection following the manufacturer’s instruction. Before that, we treated cells by 3000 µm/mL crocin and 0.75 µm/mL epirubicin. Then, the cells were fixed until the PI staining assay was performed. Next, the percentage of cells undergoing apoptosis and the cells were entered in different cell cycle phases was determined by BD FACSCalibur flow cytometer (Becton Dickinson, Canada) and analyzed by FlowJ software.
3.7. Statistical Analyses
All the experiments were carried out in triplicate, independently, and the results were declared as the mean ± SD, after analyzing by Prism software. For mean comparison of each group to control, the Student’s t test was used (P < 0.05).
4. Results
4.1. IC50 Value
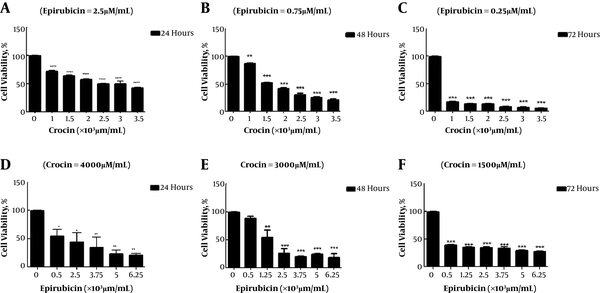
Figure 1 illustrates that crocin and epirubicin prohibit development of the OV2008 cells in a dose- and time-dependent manner at 24 hours, 48 hours, and 72 hours. The graphs illustrated that the cell viability declined by increasing hours and concentration, significantly (P < 0.05 and P < 0.0001). As we shown in Table 1, by increasing time, the IC50 of crocin and epirubicin decreased. Also, the IC50 of crocin and epirubicin combination reduced after 48 and 72 hours. According to the previously mentioned formula, the synergic index (q) was calculated for each time and the results were 0.86, 0.57, and 0.76 for 72, 48, and 24 hours, respectively. As a consequence, after 72 hours, these two components showed a simple additive effect.
IC50 Values (µm/mL) of Crocin and Epirubicin and Their Combination on OV2008 Cell Line
| Treatment, µm/mL | 24 h | 48 h | 72 h |
|---|---|---|---|
| Crocin | 5000 | 3000 | 1500 |
| Epirubicin | 2.5 | 0.75 | 0.25 |
| Crocin (constant) + epirubicin (variable) | 0.5 | 1.25 | 0.5 |
| Crocin (variable) + epirubicin (constant) | 2500 | 1500 | 1000 |
The effects of different amounts of crocin and epirubcin combination, in 24 - 48 and 72 hours on viability of OV2008 cell line (crocin variable- epirubicin constant (A - C) or vice versa (D - F)), (the results are represented as the mean ± SD of duplicate-*, **, *** P)
4.2. Apoptosis
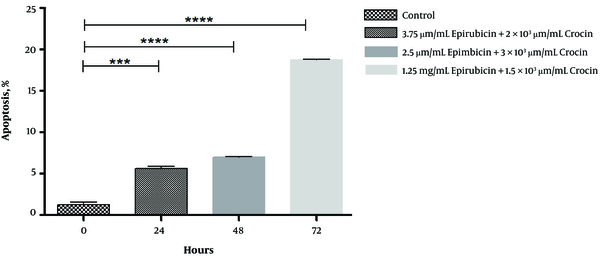
As it was revealed in Figure 2, treated cells with the lower level of epirubicin and crocin after 72 hours may lead to the most apoptosis compared to control (17%). Similarly, in comparison with the control group, the percentages of apoptotic nuclear cells were increased significantly, but around 5% after 24 and 48 hours (P = 0.0001, P < 0.0001).
Apoptotic effect of crocin and epirubicin on treated OV2008 cell line in different times which shown through Hoechst staining assay (***P = 0.0001, **** P < 0.0001)
4.3. Gene Expression
As shown in Figure 3, the selected dose of crocin and epirubicin combination diminished Bcl2 expression level in OV2008 (P < 0.05). These combined components also led to enhancing P21 and Bax genes expression significantly after 24 hours in comparison with the control group, which is more obvious in P21 (P < 0.05).
Crocin and epirubicin combination effect on mRNA expression of BAX, BCL2 and P21 in OV2008 cell line (mean ± SD, R = 2, *, **, **** P < 0.05)

4.4. Cell Cycle and Apoptosis
Table 2 enounced that after 24, 48, and 72 hours, treated cells by 3000 µm/mL crocin and 0.75 µm/mL epirubicin led to escalating dead cells percentages (subG1) in comparison with control group. Moreover, this combination affected introduced cells in each phase of the cell cycle. So, by inducing cell death, the cells which enter in the cell cycle (phase G1) and the percent of cells in the DNA replication stage (phase S) declined. Thereby, cell proliferation was suppressed after treatment.
The Amount of OV2008 Cells in 24 Hours, 48 Hours and 72 Hours Introducing in Different Phases of Cell Cycle Measured by Flowcytometrya
| Hours | Phases | |||||||
|---|---|---|---|---|---|---|---|---|
| SUB-G1 | G0/G1 | S | G2/M | |||||
| Control | Treatment | Control | Treatment | Control | Treatment | Control | Treatment | |
| 24 | 7.78 | 35.9 | 66.1 | 24.3 | 49.7 | 26.7 | 18.5 | 22.4 |
| 48 | 3.47 | 18.1 | 71.5 | 40.1 | 48 | 38 | 16.9 | 34.6 |
| 72 | 13 | 25 | 64.3 | 35.9 | 43.5 | 39 | 12.3 | 22.4 |
5. Discussion
Cervical cancer is one of the most prevalence neoplasms between women (25). In spite of the progression in therapeutic and diagnostic options, tumor cells resistance to drugs leads to recurrence of cancer (25). In order to overwhelm these complications, scholars tend to catch sight of some innovated therapy by investigating possible antitumor agent in botanies (26, 27).
In the Middle East and some European countries, saffron (Crocus sativus L.) was widely used as a condiment and a herb (28). Obviously, Crocin, as a main component of saffron, has many prominent properties such as apoptotic and anti-cancer effects, which have been demonstrated in several cancers (21, 29-32).
Apoptosis is a highly complex mechanism, which participates in chemo-resistance and done via two particular pathways: The death receptor and mitochondrial pathway. Today, they find out these pathways have interconnected with each other through some molecules (21, 33). In our survey, we demonstrated that Hoechst staining outcomes is in comprise of MTT assay and showed crocin and epirubicin combination can kill OV2008 cells. Furthermore, pursuant to our previous research (3), this aforesaid combination induced survival reduction in OV2008 cell line after 48 hours (crocin constant and epirubicin variable: IC50 = 1.25 µm/mL, crocin variable and epirubicin constant IC50 = 1500 µm/mL) and 72 hours (crocin constant and epirubicin variable: IC50 = 0.5 µm/mL, crocin variable and epirubicin constant IC50 = 1000 µm/mL).We realized that the combination of crocin and epirubicin only in 72 hours had a simple additive effect. To assay their impacts on cell cycle phases, we used flowcytometry technique. The results of the cell cycle analysis showed that this combination increased the percentage of dead cells (subG1 phase) compared to the control group and also affected the percentage of cells, which introduced in each of the cell cycle phases. We also observed that treated cells by 3000 µm/mL crocin and 0.75 µm/mL epirubicin led to increasing the percentages of secondary apoptotic and necrotic cells in comparison with control group. To further illuminate the anti-cancer mechanism of crocin on the OV2008 cell line, we encountered changes of apoptosis-related Bax, Bcl2, and P21 genes by real-time PCR technique. According to our consequences, the combination of epirubicin and crocin decreased bcl2 and increased p21 and Bax expressions over time, significantly as well as our results when we use crocin alone (3). Among many tumor suppressors, P53 plays a critical character in preventing improper cell increase and inducing cell cycle detention in the G1/G2 and S phases. P21 has been identified as a downstream target and primary intermediary of P53, follow up DNA damages (34). It also plays a key role in apoptosis and responds to stress factors by up-regulating p53 protein in order to detain cell cycle. Afterward, it caused barricading transcription of bcl2 and up-regulating Bax gene expression (35). Between Bax family proteins, Bcl2 and Bax act as important agents in the mitochondrial pathway and their ratio immovably leads to drug resistance and apoptosis capacity modulation (33, 36).
Similar studies have already been conducted. These surveys insisted that Crocin exhibited an effective inhibitory effect on proliferation of different cancer cells. For the first time, Tarantilis et al. demonstrated that crocin had an inhibitory effect on HL60 cell line growth (37). In another study, crocin and ethanolic extract of Crocus sativus exhibited a dose-dependent inhibitory effect on skin mice papillomas (38). Aung et al. also investigated the effect of crocin on human colorectal cancer cell lines (HCT-116, SW-480, HT-29) and reported a reduction in cell growth and proliferation (18). Hoshyar et al. accounted that crocin causes apoptosis by increasing bax/bcl2 ratio in AGS cell lines (human gastric adenocarcinoma cells (21). Nowadays, many studies indicated that the combination of crocin with different kinds of chemotherapy drugs may lead to enhance the efficiency of common cancer treatments through declining essential dose of these toxic medicines (32, 39-41). Meanwhile, because crocin is a safe water-soluble carotenoid, it can be used in companion with the little essential dose of chemotropic drugs such as epirubicin to get better results in cancer therapy. Nonetheless, more investigations are needed to enlighten their synergic or antagonist effects in cervical cancerous animal model.
5.1. Conclusions
Overall, the present study demonstrated the more effectiveness and stronger pro-apoptotic effect of crocin and epirubicin on OV2008 cells compare them alone through changing the expression of Bax, Bcl2, and P21 genes.
Acknowledgements
References
-
1.
Cancer Genome Atlas Research Network; Albert Einstein College of Medicine; Analytical Biological Services; Barretos Cancer Hospital; Baylor College of Medicine; Beckman Research Institute of City of Hope, et al. Integrated genomic and molecular characterization of cervical cancer. Nature. 2017;543(7645):378-84. [PubMed ID: 28112728]. [PubMed Central ID: PMC5354998]. https://doi.org/10.1038/nature21386.
-
2.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394-424. [PubMed ID: 30207593]. https://doi.org/10.3322/caac.21492.
-
3.
Mollaei H, Safaralizadeh R, Babaei E, Abedini MR, Hoshyar R. The anti-proliferative and apoptotic effects of crocin on chemosensitive and chemoresistant cervical cancer cells. Biomed Pharmacother. 2017;94:307-16. [PubMed ID: 28763753]. https://doi.org/10.1016/j.biopha.2017.07.052.
-
4.
Kooti W, Servatyari K, Behzadifar M, Asadi-Samani M, Sadeghi F, Nouri B, et al. Effective medicinal plant in cancer treatment, part 2: Review study. J Evid Based Complementary Altern Med. 2017;22(4):982-95. [PubMed ID: 28359161]. [PubMed Central ID: PMC5871268]. https://doi.org/10.1177/2156587217696927.
-
5.
Hoshyar R, Mahboob Z, Zarban A. The antioxidant and chemical properties of Berberis vulgaris and its cytotoxic effect on human breast carcinoma cells. Cytotechnology. 2016;68(4):1207-13. [PubMed ID: 25916942]. [PubMed Central ID: PMC4960168]. https://doi.org/10.1007/s10616-015-9880-y.
-
6.
Hoshyar R, Mohaghegh Z, Torabi N, Abolghasemi A. Antitumor activity of aqueous extract of Ziziphus jujube fruit in breast cancer: An in vitro and in vivo study. Asia Pac J Reprod. 2015;4(2):116-22. https://doi.org/10.1016/S2305-0500(15)30007-5.
-
7.
Hoshyar R, Mollaei H. A comprehensive review on anticancer mechanisms of the main carotenoid of saffron, crocin. J Pharm Pharmacol. 2017;69(11):1419-27. [PubMed ID: 28675431]. https://doi.org/10.1111/jphp.12776.
-
8.
Bolhassani A, Khavari A, Bathaie SZ. Saffron and natural carotenoids: Biochemical activities and anti-tumor effects. Biochim Biophys Acta. 2014;1845(1):20-30. [PubMed ID: 24269582]. https://doi.org/10.1016/j.bbcan.2013.11.001.
-
9.
Sarshoori JR, Asadi MH, Mohammadi MT. Neuroprotective effects of crocin on the histopathological alterations following brain ischemia-reperfusion injury in rat. Iran J Basic Med Sci. 2014;17(11):895-902. [PubMed ID: 25691932]. [PubMed Central ID: PMC4328099].
-
10.
Mecocci P, Tinarelli C, Schulz RJ, Polidori MC. Nutraceuticals in cognitive impairment and Alzheimer's disease. Front Pharmacol. 2014;5:147. [PubMed ID: 25002849]. [PubMed Central ID: PMC4066843]. https://doi.org/10.3389/fphar.2014.00147.
-
11.
Xiong Y, Wang J, Yu H, Zhang X, Miao C. Anti-asthma potential of crocin and its effect on MAPK signaling pathway in a murine model of allergic airway disease. Immunopharmacol Immunotoxicol. 2015;37(3):236-43. [PubMed ID: 25753844]. https://doi.org/10.3109/08923973.2015.1021356.
-
12.
Ozdemir O. Any role for probiotics in the therapy or prevention of autoimmune diseases? Up-to-date review. J Complement Integr Med. 2013;10. [PubMed ID: 23921494]. https://doi.org/10.1515/jcim-2012-0054.
-
13.
Li K, Li Y, Ma Z, Zhao J. Crocin exerts anti-inflammatory and anti-catabolic effects on rat intervertebral discs by suppressing the activation of JNK. Int J Mol Med. 2015;36(5):1291-9. [PubMed ID: 26648423]. [PubMed Central ID: PMC4601741]. https://doi.org/10.3892/ijmm.2015.2359.
-
14.
Wang Y, Li HX, Zhu LY. Experimental study of Saffron hepaotoxicity in rats. J Harbin Med Univ. 2010;2:133-5.
-
15.
Kamalipour M, Akhondzadeh S. Cardiovascular effects of saffron: An evidence-based review. J Tehran Heart Cent. 2011;6(2):59-61. [PubMed ID: 23074606]. [PubMed Central ID: PMC3466873].
-
16.
Sun Y, Xu HJ, Zhao YX, Wang LZ, Sun LR, Wang Z, et al. Crocin exhibits antitumor effects on human leukemia HL-60 cells In vitro and In vivo. Evid Based Complement Alternat Med. 2013;2013:690164. [PubMed ID: 23573146]. [PubMed Central ID: PMC3615578]. https://doi.org/10.1155/2013/690164.
-
17.
Escribano J, Alonso GL, Coca-Prados M, Fernandez JA. Crocin, safranal and picrocrocin from saffron (Crocus sativus L.) inhibit the growth of human cancer cells in vitro. Cancer Lett. 1996;100(1-2):23-30. [PubMed ID: 8620447]. https://doi.org/10.1016/0304-3835(95)04067-6.
-
18.
Aung HH, Wang CZ, Ni M, Fishbein A, Mehendale SR, Xie JT, et al. Crocin from Crocus sativus possesses significant anti-proliferation effects on human colorectal cancer cells. Exp Oncol. 2007;29(3):175-80. [PubMed ID: 18004240]. [PubMed Central ID: PMC2658895].
-
19.
Chen Y, Yang T, Huang J, Tian X, Zhao C, Cai L, et al. Comparative evaluation of the antioxidant capacity of crocetin and crocin in vivo. Chin Pharmacol Bull. 2010;26(2):248-51.
-
20.
Abedini MR, Qiu Q, Yan X, Tsang BK. Possible role of FLICE-like inhibitory protein (FLIP) in chemoresistant ovarian cancer cells in vitro. Oncogene. 2004;23(42):6997-7004. [PubMed ID: 15258564]. https://doi.org/10.1038/sj.onc.1207925.
-
21.
Hoshyar R, Bathaie SZ, Sadeghizadeh M. Crocin triggers the apoptosis through increasing the Bax/Bcl-2 ratio and caspase activation in human gastric adenocarcinoma, AGS, cells. DNA Cell Biol. 2013;32(2):50-7. [PubMed ID: 23347444]. https://doi.org/10.1089/dna.2012.1866.
-
22.
D'Alessandro AM, Mancini A, Lizzi AR, De Simone A, Marroccella CE, Gravina GL, et al. Crocus sativus stigma extract and its major constituent crocin possess significant antiproliferative properties against human prostate cancer. Nutr Cancer. 2013;65(6):930-42. [PubMed ID: 23909737]. https://doi.org/10.1080/01635581.2013.767368.
-
23.
Milajerdi A, Djafarian K, Hosseini B. The toxicity of saffron (Crocus sativus L.) and its constituents against normal and cancer cells. J Nutr Intermed Metab. 2016;3:23-32. https://doi.org/10.1016/j.jnim.2015.12.332.
-
24.
Mostafavinia SE, Khorashadizadeh M, Hoshyar R. Antiproliferative and proapoptotic effects of crocin combined with hyperthermia on human breast cancer cells. DNA Cell Biol. 2016;35(7):340-7. [PubMed ID: 27003728]. https://doi.org/10.1089/dna.2015.3208.
-
25.
Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc. 2008;3(6):1101-8. [PubMed ID: 18546601]. https://doi.org/10.1038/nprot.2008.73.
-
26.
Liao S, Xiao S, Chen H, Zhang M, Chen Z, Long Y, et al. CD38 enhances the proliferation and inhibits the apoptosis of cervical cancer cells by affecting the mitochondria functions. Mol Carcinog. 2017;56(10):2245-57. [PubMed ID: 28544069]. https://doi.org/10.1002/mc.22677.
-
27.
Mohan A, Narayanan S, Sethuraman S, Krishnan UM. Combinations of plant polyphenols & anti-cancer molecules: A novel treatment strategy for cancer chemotherapy. Anticancer Agents Med Chem. 2013;13(2):281-95. [PubMed ID: 22721388].
-
28.
Bachrach Z. Contribution of selected medicinal plants for cancer prevention and therapy. Acta Fac Med Naiss. 2012;29(3):117-23. https://doi.org/10.2478/v10283-012-0016-4.
-
29.
Abdullaev FI. Cancer chemopreventive and tumoricidal properties of saffron (Crocus sativus L.). Exp Biol Med (Maywood). 2002;227(1):20-5. [PubMed ID: 11788779]. https://doi.org/10.1177/153537020222700104.
-
30.
Garcia-Olmo DC, Riese HH, Escribano J, Ontanon J, Fernandez JA, Atienzar M, et al. Effects of long-term treatment of colon adenocarcinoma with crocin, a carotenoid from saffron (Crocus sativus L.): An experimental study in the rat. Nutr Cancer. 1999;35(2):120-6. [PubMed ID: 10693164]. https://doi.org/10.1207/S15327914NC352_4.
-
31.
Noureini SK, Wink M. Antiproliferative effects of crocin in HepG2 cells by telomerase inhibition and hTERT down-regulation. Asian Pac J Cancer Prev. 2012;13(5):2305-9. [PubMed ID: 22901211]. https://doi.org/10.7314/APJCP.2012.13.5.2305.
-
32.
Chen S, Zhao S, Wang X, Zhang L, Jiang E, Gu Y, et al. Crocin inhibits cell proliferation and enhances cisplatin and pemetrexed chemosensitivity in lung cancer cells. Transl Lung Cancer Res. 2015;4(6):775-83. [PubMed ID: 26798587]. [PubMed Central ID: PMC4700215]. https://doi.org/10.3978/j.issn.2218-6751.2015.11.03.
-
33.
Elmore S. Apoptosis: A review of programmed cell death. Toxicol Pathol. 2007;35(4):495-516. [PubMed ID: 17562483]. [PubMed Central ID: PMC2117903]. https://doi.org/10.1080/01926230701320337.
-
34.
Ashrafi M, Bathaie SZ, Abroun S, Azizian M. Effect of crocin on cell cycle regulators in n-nitroso-n-methylurea-induced breast cancer in rats. DNA Cell Biol. 2015;34(11):684-91. [PubMed ID: 26394119]. [PubMed Central ID: PMC4642822]. https://doi.org/10.1089/dna.2015.2951.
-
35.
Wang X, Wang Z, Wang J, Wang Y, Liu L, Xu X. LncRNA MEG3 has anti-activity effects of cervical cancer. Biomed Pharmacother. 2017;94:636-43. [PubMed ID: 28787698]. https://doi.org/10.1016/j.biopha.2017.07.056.
-
36.
Fulda S, Debatin KM. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene. 2006;25(34):4798-811. [PubMed ID: 16892092]. https://doi.org/10.1038/sj.onc.1209608.
-
37.
Tarantilis PA, Morjani H, Polissiou M, Manfait M. Inhibition of growth and induction of differentiation of promyelocytic leukemia (HL-60) by carotenoids from Crocus sativus L. Anticancer Res. 1994;14(5A):1913-8. [PubMed ID: 7847826].
-
38.
Konoshima T, Takasaki M, Tokuda H, Morimoto S, Tanaka H, Kawata E, et al. Crocin and crocetin derivatives inhibit skin tumour promotion in mice. Phytother Res. 1998;12(6):400-4.
-
39.
Gupta S, Jhamb B, Katiyar S. Crocin-supplemented cisplatin is highly effective in killing breast cancer cells than cisplatin alone. Cancer Res. 2014;74(19). https://doi.org/10.1158/1538-7445.AM2014-4585.
-
40.
Li X, Huang T, Jiang G, Gong W, Qian H, Zou C. Synergistic apoptotic effect of crocin and cisplatin on osteosarcoma cells via caspase induced apoptosis. Toxicol Lett. 2013;221(3):197-204. [PubMed ID: 23830991]. https://doi.org/10.1016/j.toxlet.2013.06.233.
-
41.
Vali F, Changizi V, Safa M. Synergistic Apoptotic Effect of Crocin and Paclitaxel or Crocin and Radiation on MCF-7 Cells, a Type of Breast Cancer Cell Line. Int J Breast Cancer. 2015;2015:139349. [PubMed ID: 26693354]. [PubMed Central ID: PMC4674613]. https://doi.org/10.1155/2015/139349.
