Abstract
Introduction:
Patients with immunosuppression have an increased incidence of toxoplasmosis characterized by involvement of the central nervous system. Only a few cases of toxoplasmosis associated with immunosuppressive agents have been reported. Such cases have been reported in immune suppressed patients outside the Iran, but a search of the literature has not revealed any previous reports from this country.Case Presentation:
We described a 17- year -old male, a known case of Hodgkin’s lymphoma with the diagnosis of central nervous system (CNS) toxoplasmosis.Conclusions:
As a conclusion, CNS toxoplasmosis should be considered in the differential diagnosis of immunosuppressed patients who present with neurological manifestations.Keywords
1. Introduction
Patients with malignancies who have received chemotherapy have an increased incidence of severe and diffused toxoplasmosis with involvement of the central nervous system (CNS) (1).
Patients with Hodgkin’s disease seem to be individually threatened (2) because of immunological Immunodeficiency associated with the underlying disease and the treatment modalities or both. In previous reported, toxoplasmosis have occurred after the initiation of antineoplastic therapy (3-6). The difficulty in clinical diagnosis of cerebral toxoplasmosis is related to the lack of pathognomonic clinical or radiological features (7). We describe CNS toxoplasmosis in the patient with relapsed/treated Hodgkin’s disease.
1.1. Implication
CNS toxoplasmosis should be considered in the differential diagnosis of immunosuppressed patients who present with neurological manifestations.
1.2. Ethical Considerations
This study was approved in ethics committee of Baqiyatallah University of Medical Sciences. Patient was asked to sign an informed consent form for publication. All the terms of Helsinki declaration were considered and the personal information remained anonymous.
2. Case Presentation
A 17- year -old male, a known case of Hodgkin’s lymphoma was admitted to our hospital with anorexia, diplopia, lethargy, and right leg claudication from 3 days before admission. Hodgkin’s lymphoma was in remission phase for about 7 years. Six months before admission, he was admitted to hospital because of fever, night sweats, fatigue, weight loss, splenomegaly, and multiple cervical, retroperitoneal and para-aortic lymphadenopathy that brought up a presumptive diagnosis of relapsed phase of Hodgkin’s disease. The patient was evaluated for staging and treatment. Pathological staging and biopsy of cervical lymph nodes showed stage IIIB of Hodgkin’s lymphoma recurrence. He received chemotherapy for 4 months.
On the tenth day of hospitalization, the temperature briefly rose to 39°C, and photophobia, nuchal, passive and active foot rigidity developed.
On a neurological examination, outward deviation, mydriasis, ptosis and lack of response to light were observed in the right eye. Other cranial nerves were intact. His motor strength was 5/5 in the left extremities and 4/5 in the right extremities with normal sensation. He showed dysmetria and mild hyperreflexia on the right side. Tremor on left hand with mild rigidity on left limbs were observed. Psychiatric examination also revealed depression and anxiety disorders.
A complete blood count revealed leukopenia, with white blood cell count of 2400/μL and anemia with hemoglobin level of 11.4 g/dL. Platelet count was 261000/μL and mean corpuscular volume of 84fL. Blood chemistry showed an albumin level of 2.7 g/dL, normal level of blood urea nitrogen (BUN) and creatinine. Creatine phosphokinase (CPK) level of 344 IU/L (normal 38 - 174 IU/L) and lactate dehydrogenase (LDH) level of 858 IU/L ( normal 207 - 414 IU/L). Glucose level of 126 mg/mL, aspartate aminotransferase (AST) level of 87 IU/L(normal up to 40 IU/L), alanine aminotransferase (ALT) level of 90 IU/L(normal up to 41 IU/L) and normal level of alkaline phosphatase. Ultrasound of the abdomen revealed splenomegaly.
2.1. Serological Tests Included
Wright agglutination, Widal agglutination and Coombs Wright were negative. Bacterial cultures of urine were negative. HIV antigen and antibody screening was negative. Erythrocyte sedimentation rate was 8 mm/hour and C-reactive protein (quantitative) was 3+positive. An EEG was normal.
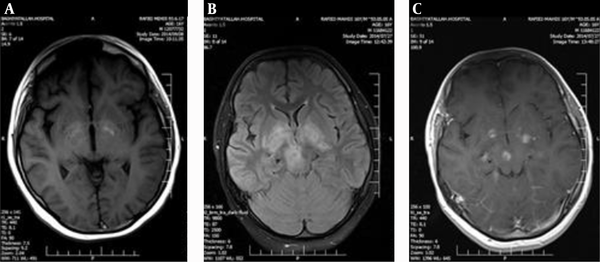
Head CT revealed hypodense lesions in the basal ganglia on the both sides, right thalamus and right cerebral peduncle.
2.2. MRI of the Brain
Axial T1W images showed patchy hyperintensity in basal ganglia and cerebral peduncles due to hemorrhage, and axial T2W and FLAIR images showed high signal intensity lesions in the basal ganglia and right cerebral peduncle, and gadolinium enhanced MRI study showed ring- like and nodular enhancing lesions in the basal ganglia and right cerebral peduncle.
According to the basal ganglia and cerebral peduncle involvement, we suspected to brain metastases arising from Hodgkin’s lymphoma so the patient was candidate for brain biopsy. But diagnosis of acute toxoplasmosis was supported by the presence of serum toxoplasma immunoglobulin M titer of 18 AU/ML (normal < 6 AU/ML) and immunoglobulin G 286 AU/ML (normal < 96 AU/ML). The result of polymerase chain reaction (PCR) examination of the cerebrospinal fluid (CSF) was positive for toxoplasma with normal level of protein, glucose and cells count.
The patient was treated with pyrimethamine (100mg loading dose orally followed by 25mg/day for the next five weeks) plus sulfadiazine (2g/day divided 4 times daily). We also added folinic acid (leucovorin) (25 mg/day) to prevent hematologic toxicity of pyrimethamine. Prednisolone 25mg per 12 hours was also ordered. A prompt therapeutic response was noted. After anti-Toxoplasma therapy initiation, progression of disease was stopped, his speech was dramatically improved but motor sequelaes remained.
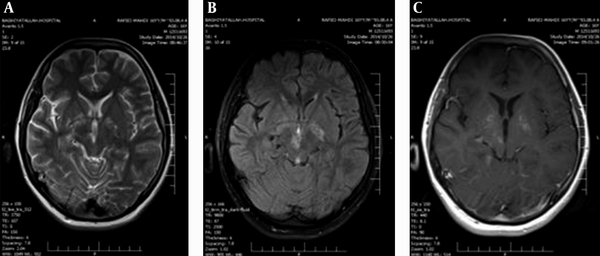
MRI of the brain at 6 weeks after anti-toxoplasmosis therapy showed dramatically regression of the lesions, typical for toxoplasmosis.
3. Discussion
Diffuse toxoplasmosis with CNS involvement is a life-threatening disease which often avoids diagnosis (1, 8). In a review of 45 patients with CNS-toxoplasmosis, less than 40% were diagnosed prior to death. However, early diagnosis of the disease has been rewarded by a rapid response to treatment (8).
Clinical reactivation is often associated with patient immunosuppression (9). Our patient was immunosuppressed because of Hodgkin’s disease and chemotherapy. The common presentation in immunosuppressed patients includes: meningoencephalitis, pneumonitis and infection (9). Focal neurological deficits are also common in immunocompromised patients (10) Some studies reported psychiatric disorders in these patients (7). Neurological deficits were of the first features of cerebral toxoplasmosis in our patient and he was also depressed and had phobia and anxiety disorders.
In a review of 85 AIDS patients with cerebral toxoplasmosis, ten (8.5%) patients did not present with any focal neurological signs. Also, the oculomotor nerve (66%) was the most frequently involved cranial nerve. In the recent case, all cranial nerves were intact.
According to centers for disease control and prevention (CDC) guidelines, criteria for establishing the diagnosis of cerebral toxoplasmosis includes the proper clinical features, consistent findings on imaging, positive serological studies, and a therapeutic response to treatment (11). All these four criteria were existed in our patient.
Only a few cases of toxoplasmosis associated with immunosuppressive agents have been reported (12-14).
The laboratory diagnosis of acute toxoplasmosis depends on serological technique (1). The most common serologic criteria are an elevated concentration of IgM Toxoplasma antibody and rise in IgG titer. In our patient, both IgM and IgG levels were increased.
CT images usually show hypodense lesions with ring enhancement or nodular enhancing lesions (15). MRI findings in CNS lymphoma included: subependymal spread and homogenous enhancement. CNS toxoplasma lesions are usually located within grey matter -white matter junction and basal ganglia and showed ring like or nodular enhancement. Also, hemorrhagic changes (TIW hyperintensity) is more likely happened in Toxoplasma rather than lymphoma.
The most frequent therapy for cerebral toxoplasmosis is pyrimethamine and sulfadiazine along with folinic acid to reduce hematological complications of pyrimethamine (15). As we used for our patient. Clindamycin is used for patients allergic to sulfa drugs (15) but it should be used in combination with pyrimethamine. The potential use of clindamycin as a single agent has not been established in randomized clinical trials (16). Roemer et al. (17) used clindamycin to treat a patient with cerebral toxoplasmosis but the patient died. While Yapar et al. (18) used clindamycin alone to treat cerebral toxoplasmosis but it took more times for complete radiological clearance of the lesions. Our patient showed good clinical improvement within 4 days and the lesion resolved approximately within 3 weeks.
A positive response to treatment can be demonstrated both clinically and radiologically. Patil et al. (19) repeated CT scan after 21 days of treatment to demonstrate response to anti-Toxoplasma therapy.
MRI of the Brain
MRI Fallowing Anti-Toxoplasma Therapy
Acknowledgements
References
-
1.
Ruskin J, Remington JS. Toxoplasmosis in the compromised host. Ann Intern Med. 1976;84(2):193-9. [PubMed ID: 766683].
-
2.
Vietzke WM, Gelderman AH, Grimley PM, Valsamis MP. Toxoplasmosis complicating malignancy. Experience at the National Cancer Institute. Cancer. 1968;21(5):816-27. [PubMed ID: 4296934].
-
3.
Gleason TH, Hamlin WB. Disseminated toxoplasmosis in the compromised host. A report of five cases. Arch Intern Med. 1974;134(6):1059-62. [PubMed ID: 4215377].
-
4.
Masson R, Fiere D, Lahneche B, Cordat C, Berger F, Revol L. [Encephalic pseudotumoral toxoplasmosis in the course of blood diseases]. Nouv Presse Med. 1975;4(35):2499-502. [PubMed ID: 1060053].
-
5.
Sarbach B. [Cerebral complications (viral encephalopathy, toxoplasmosis) in two patients with treated Hodgkin's disease. Clinical and pathological study (author's transl)]. Acta Neurol Belg. 1975;75(2):65-84. [PubMed ID: 1155022].
-
6.
Vallat M, Pesme D, Durand M, Hoerni B. [Acquired toxoplasmosis during the course of Hodgkin's disease]. Nouv Presse Med. 1976;5(38):2543-4. [PubMed ID: 980766].
-
7.
Anderson SE, Remington JS. The diagnosis of toxoplasmosis. South Med J. 1975;68(11):1433-43. [PubMed ID: 1103308].
-
8.
Townsend JJ, Wolinsky JS, Baringer JR, Johnson PC. Acquired toxoplasmosis. A neglected cause of treatable nervous system disease. Arch Neurol. 1975;32(5):335-43. [PubMed ID: 1094993].
-
9.
Halonen SK, Chiu F, Weiss LM. Effect of cytokines on growth of Toxoplasma gondii in murine astrocytes. Infect Immun. 1998;66(10):4989-93. [PubMed ID: 9746608].
-
10.
Silva LA, Vieira RS, Serafini LN, Carlotti CGJr, Figueiredo JF. [Toxoplasmosis of the central nervous system in a patient without immunosupression: case report]. Rev Soc Bras Med Trop. 2001;34(5):487-90. [PubMed ID: 11600917].
-
11.
Revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. Morb Mortal Wkly Rep. 1993;41:1-10.
-
12.
Pulivarthi S, Reshi RA, McGary CT, Gurram MK. Cerebral toxoplasmosis in a patient on methotrexate and infliximab for rheumatoid arthritis. Intern Med. 2015;54(11):1433-6. [PubMed ID: 26028002]. https://doi.org/10.2169/internalmedicine.54.3977.
-
13.
Safa G, Darrieux L. Cerebral toxoplasmosis after rituximab therapy. JAMA Intern Med. 2013;173(10):924-6. [PubMed ID: 23589123]. https://doi.org/10.1001/jamainternmed.2013.374.
-
14.
Young JD, McGwire BS. Infliximab and reactivation of cerebral toxoplasmosis. N Engl J Med. 2005;353(14):1530-1. discussion 1530-1. [PubMed ID: 16207863]. https://doi.org/10.1056/NEJMc051556.
-
15.
Pereira-Chioccola VL, Vidal JE, Su C. Toxoplasma gondii infection and cerebral toxoplasmosis in HIV-infected patients. Future Microbiol. 2009;4(10):1363-79. [PubMed ID: 19995194]. https://doi.org/10.2217/fmb.09.89.
-
16.
Madi D, Achappa B, Rao S, Ramapuram JT, Mahalingam S. Successful treatment of cerebral toxoplasmosis with clindamycin: a case report. Oman Med J. 2012;27(5):411-2. [PubMed ID: 23074553]. https://doi.org/10.5001/omj.2012.100.
-
17.
Roemer E, Blau IW, Basara N, Kiehl MG, Bischoff M, Gunzelmann S, et al. Toxoplasmosis, a severe complication in allogeneic hematopoietic stem cell transplantation: successful treatment strategies during a 5-year single-center experience. Clin Infect Dis. 2001;32(1):E1-8. [PubMed ID: 11118397]. https://doi.org/10.1086/317548.
-
18.
Yapar N, Erdenizmenli M, Oguz VA, Cakir N, Yuce A. Cerebral toxoplasmosis treated with clindamycin alone in an HIV-positive patient allergic to sulfonamides. Int J Infect Dis. 2005;9(1):64-6. [PubMed ID: 15603998]. https://doi.org/10.1016/j.ijid.2004.05.004.
-
19.
Patil HV, Patil VC, Rajmane V, Raje V. Successful treatment of cerebral toxoplasmosis with cotrimoxazole. Indian J Sex Transm Dis. 2011;32(1):44-6. [PubMed ID: 21799577]. https://doi.org/10.4103/0253-7184.81255.