Abstract
Background:
Colorectal cancer is one of the most commonly diagnosed cancers and cancer- related death worldwide. Identification of new specific biomarkers could be helpful to detection of this malignancy. Altered plasma microRNA expression has been identified in many cancers, including colorectal cancer.Objectives:
The main objective of this study was to identify the circulating microRNAs with the most expression changes in colorectal cancer patients compared with neoplasm free healthy individuals.Materials and Methods:
MicroRNA expression profiling was performed on plasma samples of 37 colorectal cancer patients and 8 normal subjects using microRNA microarray. Quantitative real-time reverse transcription polymerase chain reaction was used to validate the two selected altered microR NAs. Plasma samples from 61 colorectal cancer patients and 24 normal subjects were used in our validation study.Results:
In profiling study we found a panel of six plasma microRNAs with significant downregulation. MicroRNA-142-3p and microRNA-26a-5p were selected and validated by polymerase chain reaction. Our results demonstrated that expression levels of plasma microRNA-142-3p and microRNA-26a-5p were significantly downregulated in patients with colorectal cancer when compared to control group.Conclusions:
Our findings suggest that downregulation of plasma microRNA-142-3p and microRNA-26a-5p might serve as novel noninvasive biomarkers in the diagnosis of colorectal cancer, although more studies are needed to highlight the theoretical strengths.Keywords
1. Background
Colorectal carcinoma (CRC) is the third most malignant tumor in the world (1) with approximately 10 percent of all cancers incidence and mortality (2). It has been shown that CRC will be a preventable malignancy when precancerous lesions are diagnosed early. Despite considerable progress that has been made over the past two decades in CRC diagnostic procedures and therapeutic strategies (3-5), however still many of the CRC patients, especially those who are diagnosed in advanced stages; do not suitably respond to treatment. Thus intensive researches should be done to improve our understanding of biology, genetics and epigenetic alterations in CRC for identifying diagnostic, prognostic and predictive biomarkers and finally more effective therapeutic procedures. Recent studies have shown that expression of small RNA molecules named microRNA is significantly associated with formation and progression of CRC (6-8). MicroRNAs (miRNAs) are a class of small, non-coding, single-stranded, endogenous and highly conserved RNA molecules which post-transcriptionally modulate the expression of their target genes (9). MiRNAs can influence on colorectal carcinogenesis by direct regulation of their target mRNAs as oncogenes or tumor suppressor genes. A significant advantage of miRNAs as useful molecular tools comes from the fact that a single miRNA can modulate the expression of several mRNAs. So, for identifying the markers with greater specificities and sensitivities, working on miRNA is preferred (10). Another advantage of using the miRNA is their high stability in different physical situations and resistance to storage and handling in different biological samples (11, 12). Previous studies demonstrated that miRNAs are highly stable in serum or plasma samples with appropriate concentration (13-15). Valadi et al. in 2007 reported the secretion of miRNAs in circulation system through exosomal compartments (16). The tumor-specific expression profile of miRNAs in the body fluids as well as tumor tissues is a suitable biomarker for a variety of cancers (17-19). Moreover body fluids provide constant, non-invasive source of miRNAs as cancer biomarkers.
2. Objectives
In the present study we have compared the miRNAs expression profiles in plasma of CRC patients and healthy individuals hoping to find the miRNAs with significant altered expression in the patient group compared to the control group.
3. Materials and Methods
3.1. Subjects and Blood Samples
In this study, pre-operative plasma from 61 recently diagnosed colorectal cancer patients (including 13 stage I, 41 stage II and 7 stage III patients) and 24 (age and gender-matched) healthy subjects as the control were collected between 2012 and 2013 at Digestive Disease Research Institute (DDRI), Shariati Hospital, Tehran, Iran (Table 1). Tumors were staged according to the tumor–node–metastasis (TNM) staging system guidelines. No significant differences were observed between the CRC patients and controls in the distribution of gender (P value = 0.512, chi-square test) and age (P value = 0.8, Independent Samples t test). Normal subjects were asymptomatic individuals recruited from a colonoscopy screening. The CRC patients group consisted of 27 women and 34 men with a median age of 66 years (range, 45 - 80) and the healthy subjects consisted of 10 women and 14 men with a median age of 63 years (range, 47 - 77). The patients with a history of familial adenomatous polyposis or hereditary non-polyposis CRC or previous history of malignant tumors were excluded from this study. An amount of 5 to 10 milliliters of whole blood were obtained from each participant. The plasma was obtained by centrifugation at 1200 gr for 10 minutes at 4°C. The supernatant plasma was quickly frozen in -70°C until analysis. Informed consents were obtained from all participants for the use of their blood samples in this study. This study was approved by the Clinical Research Ethics Committee of Shariati Hospital, Tehran University of Medical Sciences.
Patient Characteristics for Plasma miRNA Analysis
| 64.13 ± 8.673 | 61.96 ± 8.67 | |
| Male | 34 | 14 |
| Female | 27 | 10 |
| I | 13 | - |
| II | 41 | - |
| III | 7 | - |
| Colon | 52 | - |
| Rectum | 9 | - |
3.2. RNA Isolation
Total RNA, including miRNA, was extracted from plasma samples by using the miRNA easy Serum/Plasma Kit (Qiagen) following the manufacturer’s instruction. For normalization of sample-to-sample variation during RNA isolation and as internal control, a synthetic miRNA (cel-miR-39 from C. elegans) was added into each denatured sample in equal amounts. RNA concentration and integrity were quantified using NanoDrop-1000 Spectrophotometer (Thermo Fisher Scientific Inc., Wilmington, DE) and Agilent’s Bioanalyzer (Agilent Technologies, Palo Alto, CA).
3.3. MiRNA Microarray Expression Profiling and Data Analysis
The miRNA expression profiles were generated by using the Agilent's miRNA Microarray system (V19), (Agilent Technologies, Santa Clara, CA), on plasma samples from 37 CRCs (including 9 stage I, 25 stage II and 3 stage III patients) and 8 normal controls. This array detects 2006 human mature miRNAs. Briefly, in labeling step 100 ng of the diluted total RNA was dephosphorylate by calf intestinal alkaline phosphatase followed by denaturation with 2.8 µL of 100% DMSO and ligation to Cyanine3-pCp using T4 RNA Ligase. After drying the samples using a vacuum concentrator and treatment with GE blocking agent and Hi-RPM hybridization buffer, samples were loaded on Agilent SureHyb chamber. In hybridization step, samples were hybridized overnight to miRNA bead chip in a hybridization oven at 55°C. After washing the samples were scanned by Agilent's Microarray Scanner. Microarray data processing was generated in the agilent feature extraction software and used for further analysis with GeneSpring GX Analysis software (Agilent Technologies Inc., USA).
3.4. Quantification of MiRNAs by qRT-PCR
100 ng of extracted total RNA was reverse transcribed with a miScript Reverse Transcription Kit (Qiagen, Valencia, CA), in a 20 μL reaction volume according to the manufacture's protocol. MiRNA expression levels were quantified in duplicate by Real-time PCR using SYBR Green miScript real-time PCR system (Qiagen, Valencia, CA) according to the manufacturer's instructions on LightCycler v.3.5 system (Roche Applied Science, Mannheim, Germany). Product amplification was performed using miRNA-specific primers and universal primers (Qiagen, Valencia, CA). In brief, the 20 μL PCR reaction mixture included 2 μL of RT product (10 ng), 10 μL 2x QuantiTect SYBR Green PCR Master Mix, PCR primer and universal primer each one 2 μL and 4 μL nuclease free water, was incubated at 95°C for 15 minutes., followed by 50 cycles of 94°C for 15 seconds, 55°C for 20 seconds and 72°C for 20 seconds. The expression levels of each miRNA were normalized against miR-16 expression and the threshold cycle (Ct) values of 40 or greater were defined as undetectable. The relative expression of plasma miRNA levels between two groups was calculated using the 2-∆∆Ct method. GraphPad Prism 6 was used for data analysis and preparing the graphs.
4. Results
4.1. MicroRNA Expression Profiling
The extracted total RNA concentration ranges were from 21.1 to 54.87 ng/μL in plasma samples of CRC patients and normal group. By data analysis of miRNA microarray on 37 CRC patients and 8 normal subjects, using precise significance criteria of a two-fold or greater difference in expression level and P value < 0.05, in GeneSpring GX software, we found that six miRNAs (miR-142-3p, miR-26a-5p, miR-27a-3p, miR-326, miR-331-3p and miR-6073) were significantly downregulated in CRC patients compared to the healthy subjects. The list of these differentially expressed miRNAs is provided in Table 2.
The Most Differentially Expressed MiRNAs in CRC Patients Versus Normal Subjects
| miR Name | MiRNAs With Low Expression in Plasma of CRC Patients | |
|---|---|---|
| P Value | Fold Change | |
| 0.029 | 11.31 | |
| 0.023 | 10.97 | |
| 0.046 | 9.58 | |
| 0.038 | 5.02 | |
| 0.033 | 9.29 | |
| 0.048 | 10.17 | |
4.2. Validation Study by qRT-PCR
Among the six differentially expressed miRNAs in our profiling study, miR-142-3p and miR-26a-5p were chosen for further qRT-PCR validation study, because they showed the most changes in expression level in miRNA microarray step. In addition, their misexpressions have been shown in different types of cancers. The expression levels of miR-142-3p and miR-26a-5p were evaluated in the plasma from 61 CRC patients and 24 healthy subjects (including the samples used for miRNA microarray analysis).
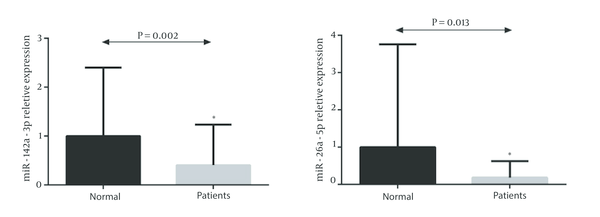
Real-time data analysis showed significant downregulation of miR-142-3p and miR-26a-5p in CRC patients compared to the normal subjects (P value = 0.002 and 0.013 respectively) (Figure 1 A and B).
Differential Expression of Circulating microrna-142-3p and MicroRNA-26a-5p in a Group of Colorectal Cancer Patients Compared to the Normal Individuals (Mann-Whitney Test)
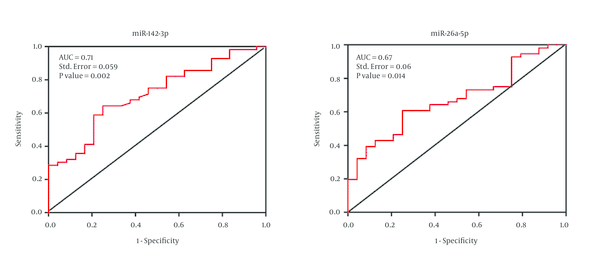
In order to evaluate the diagnostic value of the miR-142-3p and mir-26a-5p in CRC patients, ROC curve analysis was performed. These data revealed that miR-142-3p can potentially be used as a diagnostic biomarker for distinguishing between CRC patients and normal individuals. The area under the curve (AUC value) for miR-142-3p was 71% with the standard error of 0.059 (P value = 0.002) while the AUC value for miR-26a-5p was 67% with the standard error of 0.06 (P value = 0.014) (Figure 2).
Area Under Curve of Receiver Operating Characteristic for Microrna-142-3p and Microrna-26a-5p
5. Discussion
Previous studies have shown that compared to the mRNA levels, miRNA analysis may be more efficient in understanding the biology of the disease (20, 21). The main objective of this study was to identify altered expression circulating miRNAs in patients with colorectal carcinoma compared to healthy participants. Circulating miRNAs are reflective of alterations in tissue expression of miRNAs and are emerging as biomarkers in many malignancies, including colorectal cancer.
In the present study, to identify differentially expressed circulating miRNAs we have firstly compared the expression profile of miRNAs in the plasma samples from CRC patients compared to healthy controls using a miRNA microarray method and six miRNAs (miR-142-3p, miR-26a-5p, miR-27a-3p, miR-326, miR-331-3p and miR-6073) were found significantly downregulated in CRC patients. In order to validate our data, expression levels of miR-142-3p and miR-26a were evaluated in a subsequent validation study using qRT-PCR method and the results showed that the plasma levels of these two selected miRNAs were significantly decreased in CRC patients compared to the normal group.
Another downregulated miRNA, miR-326 has been already found to be downregulated in advanced breast cancer (22) and also in glioblastoma (23). Furthermore miR-326 has been identified as a potential marker associated with osseous metastasis in a lung cancer (24). MiR-331-3p expression has demonstrated to be decreased in some malignancy including breast, prostate, and pancreas cancer via targeting of human epidermal growth factor receptor 2 (HER2) (25, 26) and in glioblastoma through targeting the neuropilin-2 (NRP-2) (27). Overexpression of the long non-coding RNA HOTAIR (Hox transcript antisense intergenic RNA) has been identified in a range of tumors, including those of the colorectal cancer, hepatocellular carcinoma, pancreatic and gastric cancer, and have been shown that miR-331-3p can directly bind to HOTAIR (28). In a recent study by Wei Shen et al. miR-142-3p was found to be downregulated in colon cancer (29). They reported that reduced levels of miR-142-3p as a tumor suppressor is associated with upregulation of CD133, ATP binding cassette G2 (ABCG2), and leucine-rich-repeat-containing G-protein-coupled receptor 5 (Lgr5) by binding to their 3’-UTRs and coding sequences. Upregulation of these genes are associated with poor prognosis in colon cancer. In addition Zhou et al. found that miR-142-3p may be involved in the regulation of cell proliferation in colorectal cancer through targeting the transcription factor 7 (TCF7) (30). Another previous study revealed that miR-142-3p could regulate FASN expression which is involved in cancer cells proliferation and metastasis in CRC (31). Previously has been reported that miR-26a could functions as a potential tumor-suppressor by targeting the MYC oncogene (32, 33). Furthermore low expression levels of miR-26a has been shown in Melanoma (34), lung cancer (35), breast cancer (36) and nasopharyngeal carcinoma (33). Most recently, Jinushi et al., identified that miR-26a expression level may be used as a prognostic biomarker in CRC (37).
This research will serve as a base for future studies. Taken together our finding indicated that the differential expression of circulating miRNAs may have potential value in CRC diagnosis and might serve as novel minimally invasive molecular biomarker for this malignancy. Although a limitation of this study was the relatively small number of patients and controls and further large-scale experimental investigations are needed to verify the results of this study.
Acknowledgements
References
-
1.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225-49. [PubMed ID: 19474385]. https://doi.org/10.3322/caac.20006.
-
2.
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60(5):277-300. [PubMed ID: 20610543]. https://doi.org/10.3322/caac.20073.
-
3.
Hewitson P, Glasziou P, Watson E, Towler B, Irwig L. Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (hemoccult): an update. Am J Gastroenterol. 2008;103(6):1541-9. [PubMed ID: 18479499]. https://doi.org/10.1111/j.1572-0241.2008.01875.x.
-
4.
Schoen RE, Pinsky PF, Weissfeld JL, Yokochi LA, Church T, Laiyemo AO, et al. Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N Engl J Med. 2012;366(25):2345-57. [PubMed ID: 22612596]. https://doi.org/10.1056/NEJMoa1114635.
-
5.
Zauber AG, Winawer SJ, O'Brien MJ, Lansdorp-Vogelaar I, van Ballegooijen M, Hankey BF, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366(8):687-96. [PubMed ID: 22356322]. https://doi.org/10.1056/NEJMoa1100370.
-
6.
Srivastava K, Srivastava A. Comprehensive review of genetic association studies and meta-analyses on miRNA polymorphisms and cancer risk. PLoS One. 2012;7(11). e50966. [PubMed ID: 23226435]. https://doi.org/10.1371/journal.pone.0050966.
-
7.
Tokarz P, Blasiak J. The role of microRNA in metastatic colorectal cancer and its significance in cancer prognosis and treatment. Acta Biochim Pol. 2012;59(4):467-74. [PubMed ID: 23173124].
-
8.
Bhardwaj A, Singh S, Singh AP. MicroRNA-based Cancer Therapeutics: Big Hope from Small RNAs. Mol Cell Pharmacol. 2010;2(5):213-9. [PubMed ID: 21289871]. https://doi.org/10.4255/mcpharmacol.10.27.
-
9.
Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215-33. [PubMed ID: 19167326]. https://doi.org/10.1016/j.cell.2009.01.002.
-
10.
Visone R, Croce CM. MiRNAs and cancer. Am J Pathol. 2009;174(4):1131-8. [PubMed ID: 19264914]. https://doi.org/10.2353/ajpath.2009.080794.
-
11.
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A. 2008;105(30):10513-8. [PubMed ID: 18663219]. https://doi.org/10.1073/pnas.0804549105.
-
12.
Hui AB, Shi W, Boutros PC, Miller N, Pintilie M, Fyles T, et al. Robust global micro-RNA profiling with formalin-fixed paraffin-embedded breast cancer tissues. Lab Invest. 2009;89(5):597-606. [PubMed ID: 19290006]. https://doi.org/10.1038/labinvest.2009.12.
-
13.
Chim SS, Shing TK, Hung EC, Leung TY, Lau TK, Chiu RW, et al. Detection and characterization of placental microRNAs in maternal plasma. Clin Chem. 2008;54(3):482-90. [PubMed ID: 18218722]. https://doi.org/10.1373/clinchem.2007.097972.
-
14.
Lawrie CH, Gal S, Dunlop HM, Pushkaran B, Liggins AP, Pulford K, et al. Detection of elevated levels of tumour-associated microRNAs in serum of patients with diffuse large B-cell lymphoma. Br J Haematol. 2008;141(5):672-5. [PubMed ID: 18318758]. https://doi.org/10.1111/j.1365-2141.2008.07077.x.
-
15.
Wong TS, Liu XB, Wong BY, Ng RW, Yuen AP, Wei WI. Mature miR-184 as Potential Oncogenic microRNA of Squamous Cell Carcinoma of Tongue. Clin Cancer Res. 2008;14(9):2588-92. [PubMed ID: 18451220]. https://doi.org/10.1158/1078-0432.CCR-07-0666.
-
16.
Valadi H, Ekstrom K, Bossios A, Sjostrand M, Lee JJ, Lotvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-9. [PubMed ID: 17486113]. https://doi.org/10.1038/ncb1596.
-
17.
Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6(11):857-66. [PubMed ID: 17060945]. https://doi.org/10.1038/nrc1997.
-
18.
Calin GA, Ferracin M, Cimmino A, Di Leva G, Shimizu M, Wojcik SE, et al. A MicroRNA signature associated with prognosis and progression in chronic lymphocytic leukemia. N Engl J Med. 2005;353(17):1793-801. [PubMed ID: 16251535]. https://doi.org/10.1056/NEJMoa050995.
-
19.
Schetter AJ, Leung SY, Sohn JJ, Zanetti KA, Bowman ED, Yanaihara N, et al. MicroRNA expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. JAMA. 2008;299(4):425-36. [PubMed ID: 18230780]. https://doi.org/10.1001/jama.299.4.425.
-
20.
Xi Y, Formentini A, Chien M, Weir DB, Russo JJ, Ju J, et al. Prognostic Values of microRNAs in Colorectal Cancer. Biomark Insights. 2006;2:113-21. [PubMed ID: 18079988].
-
21.
Lu J, Getz G, Miska EA, Alvarez-Saavedra E, Lamb J, Peck D, et al. MicroRNA expression profiles classify human cancers. Nature. 2005;435(7043):834-8. [PubMed ID: 15944708]. https://doi.org/10.1038/nature03702.
-
22.
Liang Z, Wu H, Xia J, Li Y, Zhang Y, Huang K, et al. Involvement of miR-326 in chemotherapy resistance of breast cancer through modulating expression of multidrug resistance-associated protein 1. Biochem Pharmacol. 2010;79(6):817-24. [PubMed ID: 19883630]. https://doi.org/10.1016/j.bcp.2009.10.017.
-
23.
Wang S, Lu S, Geng S, Ma S, Liang Z, Jiao B. Expression and clinical significance of microRNA-326 in human glioma miR-326 expression in glioma. Med Oncol. 2013;30(1):373. [PubMed ID: 23292865]. https://doi.org/10.1007/s12032-012-0373-y.
-
24.
Valencia K, Martin-Fernandez M, Zandueta C, Ormazabal C, Martinez-Canarias S, Bandres E, et al. miR-326 associates with biochemical markers of bone turnover in lung cancer bone metastasis. Bone. 2013;52(1):532-9. [PubMed ID: 23142363]. https://doi.org/10.1016/j.bone.2012.10.033.
-
25.
Epis MR, Giles KM, Barker A, Kendrick TS, Leedman PJ. miR-331-3p regulates ERBB-2 expression and androgen receptor signaling in prostate cancer. J Biol Chem. 2009;284(37):24696-704. [PubMed ID: 19584056]. https://doi.org/10.1074/jbc.M109.030098.
-
26.
Vernimmen D, Gueders M, Pisvin S, Delvenne P, Winkler R. Different mechanisms are implicated in ERBB2 gene overexpression in breast and in other cancers. Br J Cancer. 2003;89(5):899-906. [PubMed ID: 12942124]. https://doi.org/10.1038/sj.bjc.6601200.
-
27.
Epis MR, Giles KM, Candy PA, Webster RJ, Leedman PJ. miR-331-3p regulates expression of neuropilin-2 in glioblastoma. J Neurooncol. 2014;116(1):67-75. [PubMed ID: 24142150]. https://doi.org/10.1007/s11060-013-1271-7.
-
28.
Liu XH, Sun M, Nie FQ, Ge YB, Zhang EB, Yin DD, et al. Lnc RNA HOTAIR functions as a competing endogenous RNA to regulate HER2 expression by sponging miR-331-3p in gastric cancer. Mol Cancer. 2014;13:92. [PubMed ID: 24775712]. https://doi.org/10.1186/1476-4598-13-92.
-
29.
Shen WW, Zeng Z, Zhu WX, Fu GH. MiR-142-3p functions as a tumor suppressor by targeting CD133, ABCG2, and Lgr5 in colon cancer cells. J Mol Med (Berl). 2013;91(8):989-1000. [PubMed ID: 23619912]. https://doi.org/10.1007/s00109-013-1037-x.
-
30.
Zhou JL, Jiang Z, Wang ZW, Zou ST, Zhang YX, Cai W, et al. MicroRNA-142-3p is frequently upregulated in colorectal cancer and may be involved in the regulation of cell proliferation. Chin Sci Bull. 2013;58(23):2836-45. https://doi.org/10.1007/s11434-013-5937-5.
-
31.
Zaytseva YY, Rychahou PG, Gulhati P, Elliott VA, Mustain WC, O'Connor K, et al. Inhibition of fatty acid synthase attenuates CD44-associated signaling and reduces metastasis in colorectal cancer. Cancer Res. 2012;72(6):1504-17. [PubMed ID: 22266115]. https://doi.org/10.1158/0008-5472.CAN-11-4057.
-
32.
Sander S, Bullinger L, Klapproth K, Fiedler K, Kestler HA, Barth TF, et al. MYC stimulates EZH2 expression by repression of its negative regulator miR-26a. Blood. 2008;112(10):4202-12. [PubMed ID: 18713946]. https://doi.org/10.1182/blood-2008-03-147645.
-
33.
Lu J, He ML, Wang L, Chen Y, Liu X, Dong Q, et al. MiR-26a inhibits cell growth and tumorigenesis of nasopharyngeal carcinoma through repression of EZH2. Cancer Res. 2011;71(1):225-33. [PubMed ID: 21199804]. https://doi.org/10.1158/0008-5472.CAN-10-1850.
-
34.
Reuland SN, Smith SM, Bemis LT, Goldstein NB, Almeida AR, Partyka KA, et al. MicroRNA-26a is strongly downregulated in melanoma and induces cell death through repression of silencer of death domains (SODD). J Invest Dermatol. 2013;133(5):1286-93. [PubMed ID: 23190898]. https://doi.org/10.1038/jid.2012.400.
-
35.
Dang X, Ma A, Yang L, Hu H, Zhu B, Shang D, et al. MicroRNA-26a regulates tumorigenic properties of EZH2 in human lung carcinoma cells. Cancer Genet. 2012;205(3):113-23. [PubMed ID: 22469510]. https://doi.org/10.1016/j.cancergen.2012.01.002.
-
36.
Zhang B, Liu XX, He JR, Zhou CX, Guo M, He M, et al. Pathologically decreased miR-26a antagonizes apoptosis and facilitates carcinogenesis by targeting MTDH and EZH2 in breast cancer. Carcinogenesis. 2011;32(1):2-9. [PubMed ID: 20952513]. https://doi.org/10.1093/carcin/bgq209.
-
37.
Jinushi T, Shibayama Y, Kinoshita I, Oizumi S, Jinushi M, Aota T, et al. Low expression levels of microRNA-124-5p correlated with poor prognosis in colorectal cancer via targeting of SMC4. Cancer Med. 2014;3(6):1544-52. [PubMed ID: 25081869]. https://doi.org/10.1002/cam4.309.